In adults, biologics targeting specific cytokines are effective, well-tolerated, and a safe alternative to conventional systemic treatments. At present, 3 biologics (i.e. etanercept, adalimumab, and ustekinumab) are approved in paediatric psoriasis and dupilumab is approved in AD.
TNF-blockers and ustekinumab showed similar efficacy and tolerability compared with trials in an adult population [2-4]. In the meantime, long-term data from open-label extensions with TNF-blockers showed reassuring results. After 5 years, etanercept was still well-tolerated and efficacy was maintained in those who remained in the study for up to 264 weeks [5]. There were no opportunistic infections or malignancies. Similarly, 52-week data from adalimumab showed no malignancies but nasopharyngitis and headache was reported in > 20% of patients and injection-site reactions in 3.7% [6,7]. One study included not only patients with psoriasis but also with polyarticular juvenile idiopathic arthritis, enthesitis-related arthritis, and Crohn's disease. The safety profile was generally similar across indications [7].
Real-world data confirms safety in paediatric psoriasis
Real-world studies can provide further insight into the true benefit of a therapy beyond randomised controlled trials with their strict inclusion criteria. A retrospective review was conducted on the use of systemic treatments for moderate-to-severe psoriasis in children in 20 centres in the USA and Europe. All children with moderate-to-severe psoriasis who used systemic medication or phototherapy for at least 3 months were included [8]. Mean duration of treatment was 20 months with biologics and 18.7 months with methotrexate. In this review, 270 children were treated with methotrexate and 106 with biologics (primarily etanercept). Medication-related adverse events occurred less often with biologics than with methotrexate. There were less gastrointestinal side effects, fewer laboratory issues, and less adverse events leading to treatment discontinuation, but more infections with biologic therapy (see Figure) [8].
Figure: Paediatric patients treated with biologics have less gastrointestinal side effects, lab issues, adverse events leading to treatment discontinuation than methotrexate. Yet, more infections occurred with biologics [8]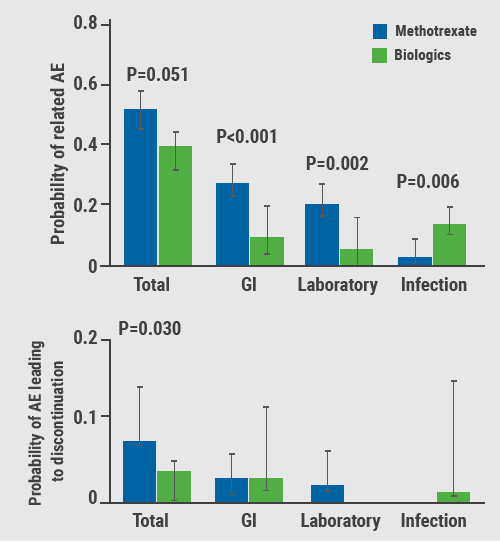 AE, adverse events; GI, gastrointestinal.
AE, adverse events; GI, gastrointestinal.
A second real-life study was performed in France, including 134 children with moderate-to-severe psoriasis in 30 centres [9]. The mean age of onset of treatment was 7.2 years and the mean age at onset of treatment with a biologic 13.2 years. Altogether, the study observed 232 years of cumulative treatment. During this period, 7 serious adverse events were documented: 3 serious infections, 2 cases of weight gain, 1 psoriasis flare, and 1 malaise [9]. Ustekinumab had the best drug survival outcome in this study. Similarly, dupilumab was equally effective and had a similar safety profile in paediatric patients with moderate-to-severe AD compared with adult patients [10]. “Basically dupilumab has the same side effects in children as in adults, including conjunctivitis,” said Prof. Marieke Seyger (Radboud University Nijmegen, the Netherlands).
Biologics are effective in both psoriasis and AD and show reassuring safety profiles. Further research and post-marketing registries are needed to collect long-term safety data of this particular sensitive patient population. “Taken together, biologics are generally associated with less treatment-related toxicity than conventional systemic agents,” concluded Prof. Seyger.
- Seyger M. D1T01.1E, EADV 2019, 9-13 Oct, Madrid, Spain.
- Paller AS, et al. N Engl J Med 2008;358:241-51.
- Papp K, et al. Lancet 2017;390:40-9.
- Landells I, et al. J Am Acad Dermatol 2015;73:594-603.
- Paller AS, et al. J Am Acad Dermatol 2016;74:280-7.
- Thaçi D, et al. Br J Dermatol 2019 Apr 24 [Epub ahead of print].
- Horneff G, et al. J Pediatr 2018;201:166-75.
- Bronckers IMGJ, et al. JAMA Dermatol 2017;153:1147-57.
- Phan C, et al. J Eur Acad Dermatol Venereol 2019;33:1984-92.
- Siegfried EC, et al. Pediatric Dermatol 2019;36:172-6.
Posted on
Previous Article
« No cancer risk with long-term use of tacrolimus, a topical calcineurin inhibitor, in children with AD Next Article
How to manage conjunctivitis in AD patients treated with a biologic »
« No cancer risk with long-term use of tacrolimus, a topical calcineurin inhibitor, in children with AD Next Article
How to manage conjunctivitis in AD patients treated with a biologic »
Table of Contents: EADV 2019
Featured articles
Late-Breaking News
IL-17A blocker effective in paediatric psoriasis patients
Rituximab beats mycophenolate mofetil in pemphigus vulgaris
Acne highly influenced by climate, pollutants, and unhealthy diet
JAK inhibition plus TCS lead to high clearance rates in AD
No cancer risk with long-term use of tacrolimus, a topical calcineurin inhibitor, in children with AD
Green light for a second JAK inhibitor in AD
Topical ruxolitinib effective in vitiligo
Emerging Therapies
Small molecules: interesting novel treatment options in AD
IL-1⍺ blockade: a new treatment option in AD
IL-4/IL-13 blockade leads to rapid itch reduction in adolescents
How to manage conjunctivitis in AD patients treated with a biologic
Biologics: increasingly used in paediatric dermatology
Spotlight on Psoriasis
IL-17 blocker: effective and safe in patients with comorbidities
ESPRIT registry: sharp decline in mortality in patients treated with a TNF blocker
Relationship psoriasis and NAFLD: new data on the hepato-dermal axis
Novel selective IL-23 blocker equally effective in patients with metabolic syndrome
Selective IL-23 blocker crushes fumaric acids in all assessed efficacy endpoints
No hint of teratogenicity through ixekizumab
New Insights in Photoprotection
Systemic photoprotection: a valuable addition to topical sun protection
The underestimated effect of visible light
Urticaria
Comorbidities more common in chronic urticaria, psoriasis, and AD
D-Dimer as future biomarker in CSU management?
Ligelizumab for CSU: symptom control and high response rates in re-treatment
Rosacea – From New Spectrum to New Therapy
New guidance on rosacea therapy according to phenotype
Best of the Posters
Above-the-neck melanoma more prone to metastases
Reduced sleep quality in dermatoses influenced by itch and pain
Anxiety and depression are common in families of AD infants
Certolizumab pegol efficacious for head and neck psoriasis
Related Articles
May 9, 2019
IBD risk of treatment with IL-17 antagonists
February 2, 2022
Psoriasis associated with a higher cancer risk
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com

