A number of studies on this subject were presented at the AAN 2019 meeting:
- A real-world study looked at discontinuation and effectiveness of dimethyl fumarate (n=428) and fingolimod (n=358) over 36 months in 786 MS patients [1]. More patients on dimethyl fumarate discontinued than on fingolimod (58.3% vs 45.2%; OR 1.81; P<0.001). The main cause was intolerance. Within 12 months of discontinuation, one third of patients had switched to high efficacy therapy (natalizumab, rituximab, ocrelizumab, alemtuzumab), while 39-48% switched to a first-line injectable or oral disease-modifying treatment. After switching to high efficacy therapy, 6% of the dimethyl fumarate group and 11.9% of the fingolimod group relapsed; after switching to an injectable/oral disease-modifying treatment, relapses were more common: 14.2% and 18.4%, respectively. There were also fewer Gd+ lesions after escalation to high efficacy therapy.
- An analysis of the PANGAEA 2.0 database revealed a benefit within 12 months of treatment and sustained effectiveness over 24 months in patients on dimethyl fumarate, teriflunomide or daclizumab who switched to fingolimod [2]. Mean reduction in annualised relapse rate at 24 months was 73.5% in the oral disease-modifying treatment group, and 72.5% in the dimethyl fumarate group.
- In a prospective, observational, multicentre study, alemtuzumab proved to be safe and effective in controlling disease activity in patients who switched from fingolimod [3]. After 12 months (n=51), the annualised relapse rate was reduced from 1.35 to 0.16 (P<0.001). The proportion of patients who developed new Gd+ lesions decreased from 59% to 3%. After 24 months (n=24), the annualised relapse rate had decreased from 1.46 to 0.16 (P<0.01). There was a reduction from 71% to 14% in the proportion of patients who developed new Gd+ lesions. Infections were detected in 55% and 52% of patients, respectively; 6% and 21% of patients developed thyroid disease.
- In the OCTAVE study, patients on ocrelizumab previously treated with natalizumab are followed. At 6 months, all 16 evaluated patients were free of relapses, EDSS progression, and MRI changes [4]. However, there were 4 serious adverse events, including 1 case each of breast cancer and acute cystitis. The authors found these possibly treatment-related events concerning, given the small number of patients.
- Hersh C, et al. AAN 2019, P3.2-065.
- Ziemssen T, et al. AAN 2019, P3.2-075.
- Gonzalez I, et al. AAN 2019, P3.2-050.
- Smoot K, et al. AAN 2019, P3.2-056.
Posted on
Previous Article
« Lasmiditan: rapid onset of efficacy in acute migraine Next Article
Long term safety of diazepam nasal spray »
« Lasmiditan: rapid onset of efficacy in acute migraine Next Article
Long term safety of diazepam nasal spray »
Table of Contents: AAN 2019
Featured articles
Letter from the Editor
Interview with Prof. Natalia Rost
Alzheimer's Disease and other Dementias
Amyloid PET in cognitively impaired patients
Tight blood pressure control lowers risk of mild cognitive impairment
Epilepsy
Headache and Migraine
Multiple Sclerosis and NMOSD
Immune tolerance by peptide-loaded tolerogenic dendritic cells
Biotin, ocrelizumab, and ibudilast in progressive MS
No increased MS relapse risk postpartum
Neuromuscular Disorders
First-ever effective and safe treatment of CMT1A
Parkinson’s Disease and other Movement Disorders
Leukaemia and hypertension therapies tested in Parkinson’s disease
Stroke
Miscellaneous
Possibly lifesaving therapy in refractory PML
New AAN guideline for treating Tourette syndrome
Subspecialty teleneurology: feasible and highly valued
Related Articles
July 30, 2019
Two experimental antibodies reduce amyloid levels
July 30, 2019
Brain-responsive neurostimulation reduces seizures
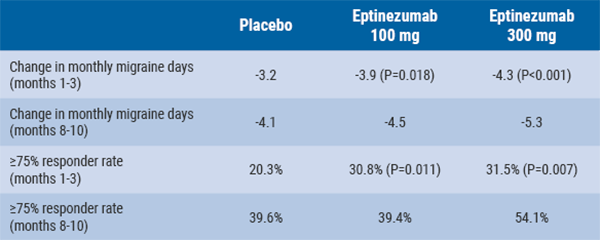
July 30, 2019
Eptinezumab reduces mean monthly migraine days
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com

