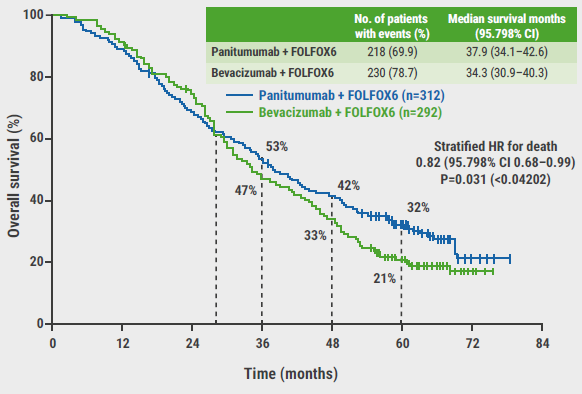
https://doi.org/10.55788/4e9ad1c3
Treatment of locally advanced rectal cancer with chemoradiotherapy followed by surgery and subsequent adjuvant chemotherapy controls local disease, but distant metastases remain common. Previously, both the RAPIDO (NCT01558921) and PRODIGE 23 (NCT01804790) trials reported better short-term outcomes (pCR rate of about 26%) and higher metastasis-free survival (MFS) using induction (PRODIGE 23) or consolidation (RAPIDO) chemotherapy while no significant 3-year OS improvement was observed [1–3]. Prof. Thierry Conroy (Institut de Cancérologie de Lorraine, France) presented 7-year results from PRODIGE 23 [4].
PRODIGE 23 enrolled 461 patients with locally advanced rectal cancer (cT3 or cT4, Nx, M0) who were randomised 1:1 to standard-of-care (i.e. neoadjuvant chemoradiotherapy followed by surgery and 12 cycles of adjuvant mFOLFIRINOX) or total neoadjuvant therapy (TNT; 6 cycles of neoadjuvant mFOLFIRINOX followed by neoadjuvant chemoradiotherapy, surgery, and 6 cycles of adjuvant mFOLFIRINOX). The primary endpoint was disease-free survival (DFS). Key secondary endpoints were MFS and OS. Restricted median survival time (RMST) at 7 years was chosen in the statistical analyses.
At 7 years, the cumulative incidence of local cancer recurrence was 5.3% versus 8.1% in the TNT arm versus standard-of-care arm, respectively. The cumulative incidence of metastatic disease was 20.7% versus 27.7%, respectively. The number of deaths were 18.2% versus 24.4%, respectively. The 7-year DFS rate was 67.6% versus 62.5%, resulting in an RMST of 5.73 months of DFS benefit for the TNT arm (P=0.048).
The secondary endpoint of 7-year MFS was 73.6% versus 65.4%, respectively, resulting in an RMST of 7.1 months of MFS benefit for the TNT arm (1 month of MFS benefit/year; P=0.011). The 7-year OS was 81.9% versus 76.1%, respectively, resulting in an RMST of 4.37 months benefit for the TNT arm (P=0.033; see Figure).
Figure: 7-year overall survival in the PRODIGE 23 trial [1]
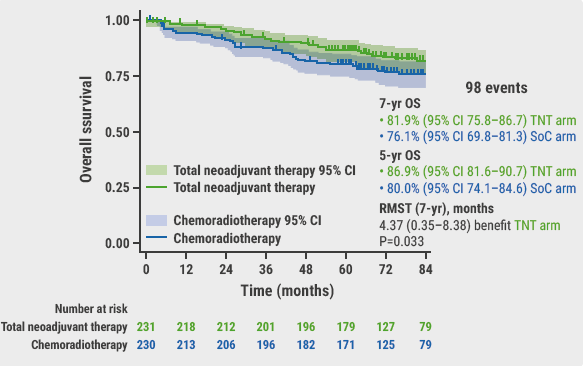
CI, confidence interval; OS, overall survival; RMST, restricted median survival time; SoC, standard-of-care; TNT, total neoadjuvant therapy.
“These long-term outcomes demonstrated that induction chemotherapy with mFOLFIRINOX before neoadjuvant chemoradiotherapy improves OS of patients with locally advanced rectal cancer. Therefore, TNT with neoadjuvant mFOLFIRINOX should now be considered as one of the best options of care for these patients,” Prof. Conroy concluded.
- Bahadoer RR, et al. Lancet Oncol. 2021;22:29–42.
- Conroy T, et al. Lancet Oncol. 2021;22:702–715.
- Bascoul-Molevi C, et al. Eur J Cancer. 2023;186:151–165.
- Conroy T, et al. Total neoadjuvant therapy with mFOLFIRINOX versus preoperative chemoradiation in patients with locally advanced rectal cancer: 7-year results of PRODIGE 23 phase III trial, a UNICANCER GI trial. Abstract LBA3504, ASCO Annual Meeting 2023, 2–6 June, Chicago, USA.
Copyright ©2023 Medicom Medical Publishers
Posted on
Previous Article
« Neoadjuvant chemotherapy may be viable option in locally advanced colon cancer Next Article
Letter from the Editor »
« Neoadjuvant chemotherapy may be viable option in locally advanced colon cancer Next Article
Letter from the Editor »
Table of Contents: ASCO 2023
Featured articles
Real-world data support new SOC in patients with SCLC
What can real-world evidence teach us about atezolizumab plus bevacizumab in HCC?
Colorectal Cancer
7-year outcomes of PRODIGE 23 trial
Neoadjuvant chemotherapy may be viable option in locally advanced colon cancer
De-escalation of neoadjuvant treatment of locally advanced rectal cancer is non-inferior
Breast Cancer
SONIA: No survival benefit with first-line versus second-line CDK4/6 inhibition in metastatic breast cancer
Adjuvant ribociclib improves invasive DFS in early breast cancer
Gene expression profiles predict benefit of neoadjuvant immune checkpoint therapy in triple-negative breast cancer
Lung Cancer
Adding pembrolizumab to perioperative chemotherapy improves EFS in early-stage NSCLC
TTFields therapy: a new treatment modality for metastatic NSCLC
Adding chemotherapy to EGFR TKI does not improve OS in advanced EGFR-mutated NSCLC
Upper GI Cancer
No improved OS in pancreatic cancer after neoadjuvant mFOLFIRINOX
AI detects gastric cancer with high accuracy in common blood tests
Melanoma
Response-directed treatment personalisation in stage III melanoma
Prognostic and predictive biomarkers in patients with resected stage IIB/C melanoma
GU Cancers
Combining PARP inhibition and androgen receptor-signalling inhibition improves radiographic progression-free survival in HRR-deficient mCRPC
Erdafitinib outperforms chemotherapy in FGFR-altered advanced urothelial cancer
Probiotic CBM588 seems to improve clinical effect cabozantinib/nivolumab in mRCC
Exploratory analysis of IMvigor130 trial finds no OS benefit from atezolizumab in subgroups
Miscellaneous
Immune checkpoint inhibition improves PFS in non-BRCA-mutated ovarian cancer
First-line nivolumab-AVD improves PFS both in adult and paediatric patients with advanced Hodgkin lymphoma
Vorasidenib successfully targets IDH1/2-mutated glioma
ASCO Interviews
IMbrave050: Adjuvant atezolizumab plus bevacizumab provides landmark recurrence-free survival for HCC
What can real-world evidence teach us about atezolizumab plus bevacizumab in HCC?
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy