https://doi.org/10.55788/09f8ffe5
An overview of the insights that have been gathered over the past 2 years on COVID-19 in patients with MS [1,2] was provided by Prof. Céline Louapre (Sorbonne University, France) [3]. At the start of the pandemic, there were some key questions to be answered for MS patients:
- How severe is COVID-19 in patients with MS?
- What are the risk factors for developing a severe form of COVID-19 in patients with MS?
- Does COVID-19 modify the course of MS?
- Could COVID-19 change MS incidence?
- Does vaccination effectively prevent COVID-19 in MS patients?
An observational study in 347 MS patients who got COVID-19 between March and May 2020, found that 73 (21%) were hospitalised and 12 (3.5%) had died [4]. Age, Expanded Disability Status Scale (EDSS), and obesity were independent risk factors for severe COVID-19. Most patients who died were in the group which did not receive any MS treatment. Neurological disability strongly predicted severe COVID-19: an EDSS of 6 or more was associated with an odds ratio (OR) of severe outcome of 6.33. A pooled analysis of 1,787 symptomatic COVID-19 patients with MS showed that around 12% were hospitalised and 2.7% were admitted to the ICU or died [5]. Anti-CD20 medication (OR 2.05) and methylprednisolone (OR 2.71) were associated with severe outcome of COVID-19 (P<0.001).
To further specify these outcomes and put them into a better perspective, Prof. Louapre went on to present very recent, unpublished results of 2,709 patients in the COVISEP registry (NCT04355611). Data cut-off was in July 2022, at which point the group had a mean age of 44 years, a mean EDSS of 2.0, and included 390 secondary progressive MS patients and 208 primary progressive MS patients. In total, 251 patients (9.3%) were hospitalised and 45 (1.7%) were admitted to the ICU or died. Hospitalisation rate was around 15% until the fifth wave of COVID-19, mainly caused by the Omicron variant, after which hospitalisation percentage dropped. Before the Omicron wave, COVID-19 severity was associated with EDSS and with anti-CD20 medication, which was still the case during the Omicron wave, despite the lower number of patients (see Figure). Among vaccinated MS patients (>2 injections) only anti-CD20 medication was associated with COVID-19 severity, with an OR of 8.2 (95% CI 2.8–24.1) for severe COVID-19.
Figure: COVID-19 severity associated with EDSS and anti-CD20 medication [3]
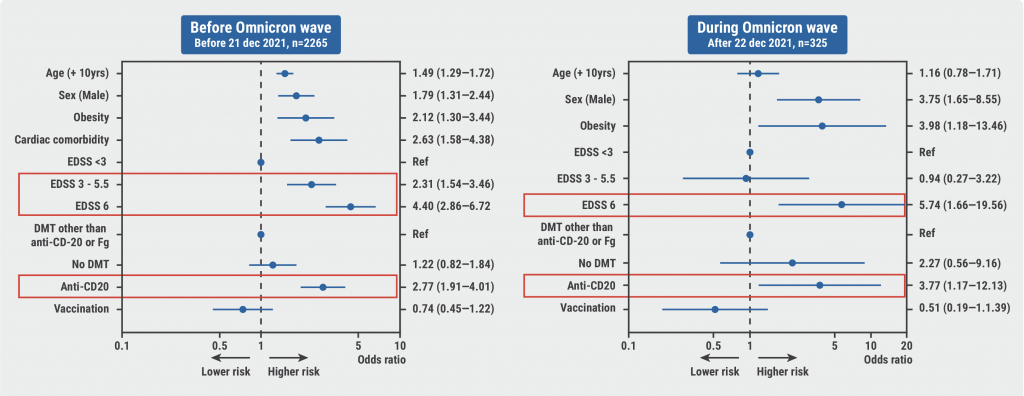
EDSS, Expanded Disability Status Scale.
To investigate whether EDSS and anti-CD20 medication are cumulative risk factors for severe COVID-19, Prof. Louapre and colleagues set up a post-hoc study including 1,005 relapsing-remitting MS patients on high-efficacy DMTs: 424 on anti-CD20 therapy and 581, with very similar characteristics, on another high-efficacy therapy (natalizumab or fingolimod). The rate of severe COVID-19 in both groups was significantly different: 13.1% and 2.6%, respectively. Applying a logistic regression model weighted by propensity scores still revealed a higher risk of severe COVID-19 in the anti-CD20 group (OR 5.02; 95% CI 2.71–9.30; P<0.001), present in all subgroups. However, this deleterious effect of anti-CD20 therapy was absent in patients with progressive MS, with an adjusted OR of 1.21 (95% CI 0.70–2.70).
So, in patients with progressive MS, the risk-benefit ratio was different: an increased risk of severe COVID-19 in patients with higher disability must be weighed against the modest efficacy of anti-CD20 therapy or in fact any treatment at this stage of the disease.
- Pugliatti M, et al. Curr Opin Neurol. 2022;35(3):319–327.
- Pugliatti M, et al. Front Immunol. 2022;13:1045101.
- Louapre C. Looking back, what have we learnt? Abstract O136, ECTRIMS 2022, 26–28 October, Amsterdam, the Netherlands.
- Louapre C, et al. JAMA Neurol. 2020;77(9):1079–88.
- Sormani MP, et al. Ann Clin Transl Neurol. 2021;8(8):1738–44.
Copyright ©2022 Medicom Medical Publishers
Posted on
Previous Article
« Ocrelizumab and fingolimod increase the risk of COVID-19 and of worse outcomes Next Article
Cognitive rehab and mindfulness reduce cognitive complaints in MS »
« Ocrelizumab and fingolimod increase the risk of COVID-19 and of worse outcomes Next Article
Cognitive rehab and mindfulness reduce cognitive complaints in MS »
Table of Contents: ECTRIMS 2022
Featured articles
Letter from the Editor
Diagnosis and Prediction of Disease Course
A case for including optic nerve lesions in the McDonald criteria
Cerebrospinal fluid kappa-free light chains for MS diagnosis
Early, non-disabling relapses increase disability accumulation
Physical impairment is present before perceived MS onset
Chronic active MS lesions respond poorly to anti-CD20 antibodies
Treatment: Trials & Strategies
Dimethyl fumarate reduces the risk of a first clinical event in RIS
How and when to make a timely switch to high-efficacy DMT
Comparing real-world effectiveness of DMTs
Study fails to show non-inferiority of rituximab to ocrelizumab
Autologous haematopoietic stem cell transplantation versus DMTs
Progressive MS
Stem cell transplantation not superior to natalizumab in progressive MS
Efficacy of DMTs fades away in secondary progressive MS
Smartphone tapping can help detect progressive MS
Paediatric MS
Early treatment with DMT effective in paediatric-onset MS
Fingolimod in paediatric MS: results of up to 6 years
Switching treatment after initial platform injectable DMT: real-world data
Pregnancy
Pregnancy and infant outcomes in women receiving ocrelizumab
New safety data of anti-CD20 mAbs around pregnancy
MS activity and pregnancy outcomes after long-term use of natalizumab
NMOSD
Ravulizumab significantly reduced relapses in AQP4+ NMOSD
NMOSD patients are cognitively impaired regardless of serostatus
Evidence-based consensus on pregnancy in NMOSD
COVID-19
COVID-19 and MS: lessons learned thus far
Ocrelizumab and fingolimod increase the risk of COVID-19 and of worse outcomes
Humoral and cellular immune responses after SARS-CoV-2 vaccination
Miscellaneous
Re-myelination strategies in MS still pose many unanswered questions
MS associated with a broader Epstein-Barr virus specific T-cell receptor repertoire
Cognitive rehab and mindfulness reduce cognitive complaints in MS
Related Articles
August 18, 2021
Positive results from ublituximab versus teriflunomide
November 25, 2020
Long-term safety and efficacy of ozanimod in RMS
January 7, 2021
Distinct cognitive phenotypes in multiple sclerosis proposed
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com

