Responders to risankizumab, an IL-23A antibody, in a phase 2 induction/maintenance study could enrol in an open-label extension (OLE). They received open-label 180 mg subcutaneous risankizumab every 8 weeks for up to 206 weeks. There were 65 CD patients who enrolled in the OLE, with 4 patients re-induced; 60 patients (92%) had been previously treated with TNF antagonists. In the OLE, median exposure to risankizumab was 1,014 (range: 114–1,317) days.
At baseline of the current study, 47 patients (72%) had clinical remission, defined as Crohn’s disease Activity Index (CDAI) <150. At baseline, 27 patients (42%) were in endoscopic remission, defined as Crohn’s disease Endoscopic Index of Severity (CDEIS) ≤4 or CDEIS ≤2 for patients with isolated ileitis at baseline. Both clinical remission and endoscopic remission were sustained up to week 152 (see Table).
Table. Clinical and endoscopic remission in patients receiving open-label risankizumab maintenance treatment [1]
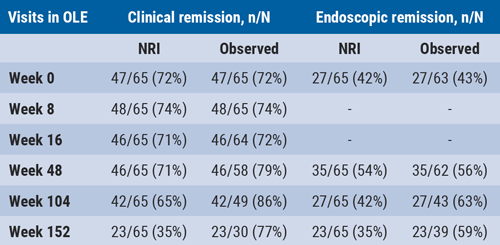
NRI, non-responder imputation.
A third of the patients (n=21) prematurely discontinued risankizumab, including 6 (9%) who experienced an adverse event (AE). Sixty patients (92%) reported AEs; in 23 (35%) these were serious. The most common AEs were nasopharyngitis (31%), gastroenteritis (23%), and fatigue (20%). Serious infections were reported in 6 patients (9%) and opportunistic infections in 3 patients (5%).
- Ferrante M, et al. ECCO-IBD 2020, OP27.
Posted on
Previous Article
« Clinical remission after dose escalation of upadacitinib Next Article
Phase 2 results of first-in-class TL1A inhibitor »
« Clinical remission after dose escalation of upadacitinib Next Article
Phase 2 results of first-in-class TL1A inhibitor »
Table of Contents: ECCO 2020
Featured articles
Gut Microbiome as Treatment Target
Response to faecal microbiota transplantation in UC
Bioactives produced by gut bacteria to modulate immune response
Big Data Analysis
Multi-omics help describe CD phenotypes
The positive impact of genetic data on drug development
Experimental Therapies: Study Results
AMT-101: an oral human IL-10 fusion protein
Phase 2 results of first-in-class TL1A inhibitor
Open-label extension study of risankizumab: final results
Clinical remission after dose escalation of upadacitinib
Short- and Long-Term Treatment Results
Infliximab discontinuation increases relapse risk
Tofacitinib ‘real-world’ effectiveness in active UC
Subcutaneous ustekinumab as maintenance therapy in UC
Subcutaneous vedolizumab maintenance therapy in CD
Vedolizumab treatment persistence and safety
Specific Therapeutic Strategies
Impact of strategies on intestinal resection rate
Early ileocaecal resection in CD patients failing conventional treatment
Biologics before surgery in IBD do not elevate infection risk
Top-down infliximab superior to step-up in children with CD
High versus standard adalimumab in active UC
Head-to-Head Comparison of Treatments
Vedolizumab and anti-TNF therapies: a real-world comparison
Cancer Risk
Increased risk of small bowel cancer in IBD
Increased incidence of colorectal cancer and death in CD
Risk of rectal, anal cancer increased in perianal CD
Glyco-fingerprint as risk factor of UC-associated cancer
Miscellaneous Topics
Resolution of mucosal inflammation has dramatic effect
PICaSSO validated in real-life study
Re-inducing inflammation in organoids from UC patients
Role of immune cells in intestinal fibrosis
Association between meat consumption and IBD risk
CD exclusion diet corrects dysbiosis
Related Articles

April 14, 2020
Impact of biologicals on faecal microbiota

April 14, 2020
Letter from the Editor
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com

