The EXCEED trial was a phase 3b study of patients with active psoriatic arthritis and an inadequate response or intolerance to conventional disease-modifying anti-rheumatic drugs (DMARDs). The trial compared the efficacy and safety of secukinumab and adalimumab as first-line biologic monotherapy through 52 weeks, with a musculoskeletal primary endpoint. The results were presented by Prof. Iain McInnes (University of Glasgow, United Kingdom), and simultaneously published in The Lancet [1,2].
Participants (n=853) were randomised to secukinumab 300 mg/week at weeks 0-4 and every 4 weeks thereafter (n=426) or adalimumab 40 mg subcutaneous at baseline and then every 2 weeks until week 50 (n=427). Baseline demographics and disease characteristics were comparable between treatment groups with the exception of higher proportion of female patients and patients without enthesitis in the secukinumab group.
At week 52, American College of Rheumatology (ACR)20 response was achieved by 67.4% of secukinumab-treated patients versus 61.5% of adalimumab-treated patients (P=0.0719). As the primary endpoint was not met, key secondary endpoints were not formally evaluated for statistical significance. However, it was clear that a Psoriasis Area and Severity Index (PASI)90 skin response was achieved by more secukinumab-treated patients than those treated with adalimumab (65% vs 43%, respectively) (see Table). Another difference between the groups concerned treatment discontinuation rates (14% with secukinumab vs 24% with adalimumab). The most important reasons for discontinuation were patient or guardian decision, adverse events, and lack of efficacy [1].
Table: Efficacy outcomes at week 52 [1]
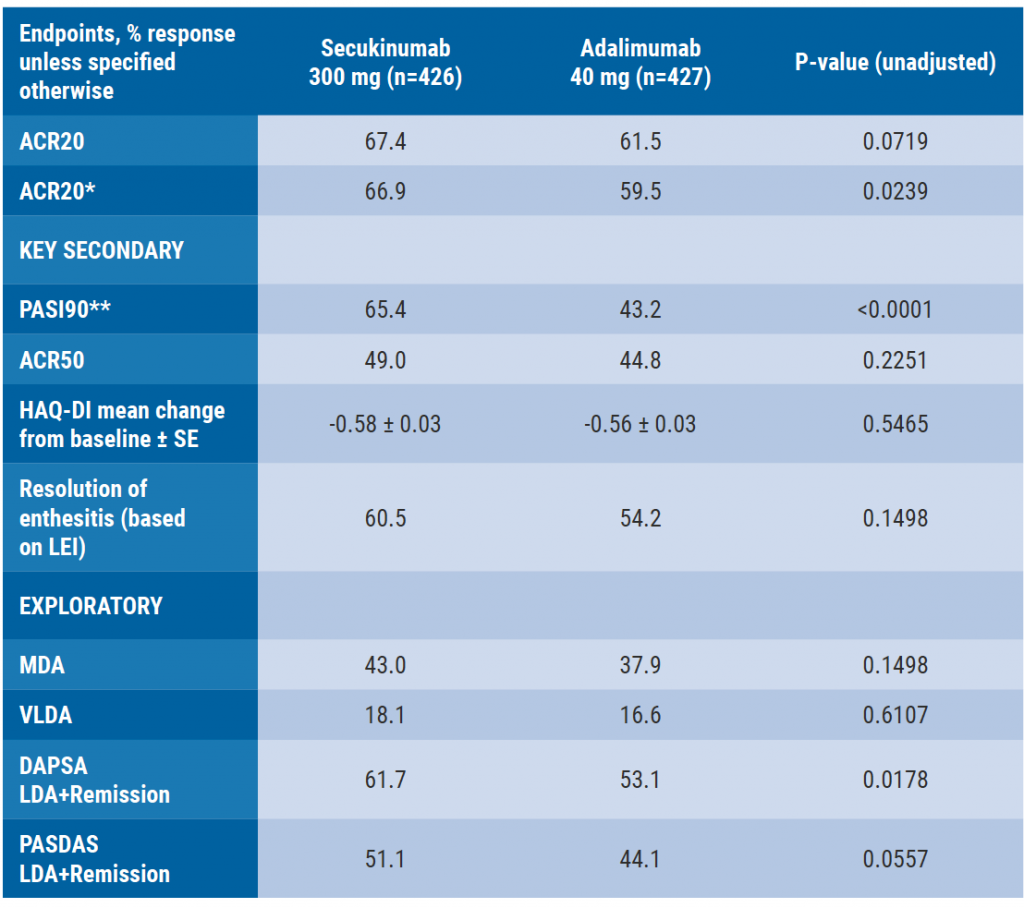
Binary variables were analysed using logistic regression. Patients who discontinued study treatment prematurely or took csDMARDs after week-36 were considered non-responders. Multiple imputation was used for all other missing data. HAQ-DI mean change from baseline was analysed using mixed-effect model repeated measures.
*Non-responder imputation was used for pre-specified sensitivity analysis
**n=215 in secukinumab and n=202 in adalimumab in psoriasis subset
***n=234 in secukinumab and n=264 in adalimumab in enthesitis subset
Furthermore, a pre-specified subgroup analysis of the trial, presented by Prof. Alice Gottlieb (Mount Sinai Hospital, New York, USA), showed that a higher proportion of patients achieved improvement in combined ACR50 and PASI100 response with secukinumab versus adalimumab (30.7% vs 19.2%, respectively; P=0.0087),adalimumab, as well as in skin specific endpoints (i.e. PASI100 and PASI score ≤3) at week 52 [3]. Higher efficacy was demonstrated for secukinumab compared with adalimumab for PASI100 responses and for the proportion of patients who achieved absolute PASI score ≤3.
- McInnes IB, et al. OP0227. EULAR E-Congress, 3-6 June 2020.
- McInnes IB, et al. The Lancet 395;10235:1496-1505.
- Gottlieb AB, et al. Abstract 0340. EULAR E-Congress, 3-6 June 2020.
Posted on
Previous Article
« Reduced maintenance dose of certolizumab pegol can be used in axSpA Next Article
Upadacitinib provides fast onset of improvement in psoriatic arthritis »
« Reduced maintenance dose of certolizumab pegol can be used in axSpA Next Article
Upadacitinib provides fast onset of improvement in psoriatic arthritis »
Table of Contents: EULAR 2020
Featured articles
COVID-19 and inflammatory rheumatic disease: some key issues
Secukinumab monotherapy as efficient as adalimumab
AxSpA real-life remission rates higher on biologics
Olokizumab significantly improves RA features and patient-reported outcomes
Rheumatoid Arthritis
New nanoparticle promising future agent in RA
Olokizumab significantly improves RA features and patient-reported outcomes
Low DAS at 4 months predicts sustained DMARD-free remission
Ankylosing Spondylitis
Reduced maintenance dose of certolizumab pegol can be used in axSpA
Worse response axSpA patients to second TNFi versus first TNFi
AxSpA real-life remission rates higher on biologics
Certolizumab pegol reduces acute anterior uveitis in axial spondyloarthritis
TNF-α inhibitors improve bone mineral density in AS patients
Psoriatic Arthritis
Ixekizumab shows sustained improvements in pain and fatigue at 3 years
Adalimumab added to methotrexate yields better results in PsA than methotrexate escalatio
Upadacitinib provides fast onset of improvement in psoriatic arthritis
Secukinumab monotherapy as efficient as adalimumab
Osteoporosis and Osteoarthritis
Higher mortality risk with tramadol versus NSAIDs for osteoarthritis patients
Hydroxychloroquine not effective in patients with hand osteoarthritis
Positive effect denosumab on fall risk
Systemic Sclerosis and Systemic Lupus Erythematosus
Anifrolumab achieves rapid and durable BICLA-response
Subclinical myocardial involvement progresses in SSc patients
Composite endpoint CRESS for primary Sjögren’s syndrome
COVID-19
COVID-19 and inflammatory rheumatic disease: some key issues
Related Articles
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com

