https://doi.org/10.55788/42cf29d6
CCA is a rare malignancy with a dismal prognosis; the median overall survival with first-line chemotherapy is approximately 1 year [1]. FGFR2 fusions or rearrangements drive 10–15% of intrahepatic CCR [2]. RLY-4008 is a highly selective FGFR2 inhibitor (FGFR2i) and has potent in-vivo activity against FGFR2 inhibitor-sensitive and resistant CCR [3]. The phase 1/2 REFOCUS trial (NCT04526106) enrolled patients with advanced solid tumours harbouring an FGFR2 alteration who received RLY-4008. Prof. Antoine Hollebecque (Institut Gustave Roussy, France) reported the results [4].
The successful phase 1 of the REFOCUS trial resulted in a recommended phase 2 dose of 70 mg every day. 38 patients with FGFR inhibitor-naïve CCA were treated with RLY-4008 in phase 1 (n=21) or phase 2 (n=17). Key objectives were investigator-assessed overall response rate (ORR) per RECIST v1.1, duration of response (DOR), and safety. The radiographic ORR was 63.2% (all partial responses). The radiographic ORR in patients treated with 70 mg was 88.2% (all partial responses; see Figure). The median time to response was 1.8 months.
The most common treatment-related adverse events (all grades) were stomatitis (42%), nail toxicities (43%), palmar-plantar erythrodysesthesia syndrome (35%), and alopecia (26%). The most common grade ≥3 treatment-related adverse event was stomatitis (8%). Interruption of treatment occurred in 42% of patients, reduction in 27% of patients, and discontinuation in 1% of patients.
Based on these results, Prof. Hollebecque resolved that “RLY-4008 is the first highly selective, irreversible inhibitor designed to target oncogenic FGFR2 driver alterations and resistance mutations. REFOCUS validates this novel mode of action and supports expedited development for treating patients with FGFR inhibitor-naïve CCA harbouring an FGFR2 fusion or rearrangement.”
- Valle J, et al. N Engl J Med. 2010;362:1273–1281.
- Jusakul A, et al. Cancer Discov. 2017;7(10):1116–1135.
- Casalleto J, et al. Cancer Res. 2021;81(13_Supplement):1455.
- Hollebecque A, et al. Efficacy of RLY-4008, a highly selective FGFR2 inhibitor in patients (pts) with an FGFR2-fusion or rearrangement (f/r), FGFR inhibitor (FGFRi)-naïve cholangiocarcinoma (CCA): ReFocus trial. Abstract LBA12, ESMO Congress 2022, 09–13 September, Paris, France.
Figure: Radiographic tumour regression and response upon RLY-4008 administration to patients with advanced solid tumours harbouring an FGFR2 alteration [4].
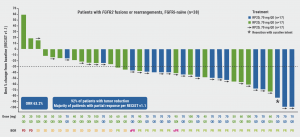
RPD2, recommended phase 2 dose. QD, once daily dosing. BOR, best overall response. PR, partial response. PD, progressive disease. SD, stable disease. uPR, unconfirmed partial response.
Copyright ©2022 Medicom Medical Publishers
Posted on
Previous Article
« Chemo-immunotherapy in gastric cancer is more effective when administered in parallel Next Article
Addition of pembrolizumab to lenvatinib does not improve OS in advanced HCC »
« Chemo-immunotherapy in gastric cancer is more effective when administered in parallel Next Article
Addition of pembrolizumab to lenvatinib does not improve OS in advanced HCC »
Table of Contents: ESMO 2022
Featured articles
Letter from the Editor
Colorectal Cancer
High pathological responses to neoadjuvant immune checkpoint inhibition in locally advanced dMMR colon cancer
Fruquintinib: a potential new treatment for patients with refractory mCRC
Second-line avelumab is effective in patients with MSI-H/dMMR mCRC
Upper Gastrointestinal Cancer
Deep learning models predict the risk of relapse and the mutational profile in GIST
Addition of pembrolizumab to lenvatinib does not improve OS in advanced HCC
New, highly selective inhibitor of FGFR2 driver alterations and resistance mutations
Chemo-immunotherapy in gastric cancer is more effective when administered in parallel
Breast Cancer
Tumour infiltrating lymphocytes identify patients with immunogenic triple-negative breast cancer
OS benefit of abemaciclib in HR-positive/HER2-negative advanced breast cancer not (yet) statistically significant
OS benefit of sacituzumab govitecan in pre-treated HR-positive/HER2-negative metastatic breast cancer
Lung Cancer
A pathway from air pollution to lung cancer in non-smokers identified
Selective KRASG12C inhibitor sotorasib demonstrates superior PFS and ORR compared to docetaxel in previously treated patients with NSCLC
Promising clinical activity of tepotinib plus osimertinib in NSCLC with MET amplification after progression on first-line osimertinib
High pathological responses in borderline resectable NSCLC patients after induction with dual immunotherapy and concurrent chemoradiotherapy
Melanoma
Treatment with tumour-infiltrating lymphocytes for advanced melanoma outperforms ipilimumab
Neoadjuvant pembrolizumab outperforms adjuvant pembrolizumab in resectable stage III–IV melanomas
Survival-benefit of neoadjuvant T-VEC maintained over 5 years of follow-up
Baseline ctDNA predicts survival in resected stage III–IV melanoma
Genitourinary Cancer – Prostate Cancer
Overall survival benefit of abiraterone in mHSPC is maintained for 7 years
Limited benefit of adding long-term ADT to post-operative radiotherapy in prostate cancer
Intensified ADT benefits biochemical progression-free survival in biochemically relapsed prostate cancer
Genitourinary Cancer – Non-Prostate Cancer
Adjuvant nivolumab plus ipilimumab does not improve survival in patients with localised RCC at high risk of relapse after nephrectomy
Triple therapy improves progression-free survival in patients with advanced RCC versus dual therapy
Adjuvant atezolizumab does not improve outcomes for patients with RCC and increased risk of recurrence
Gynaecological cancers
OS benefit for advanced ovarian cancer patients treated with maintenance olaparib
Maintenance tegafur-uracil does not improve survival in locally advanced cervical cancer
Head and Neck Cancer
Adding first-line pembrolizumab to CRT in locally advanced HNSCC does not significantly prolong survival or event-free survival
5-FU-free chemotherapy combination as an alternative for first-line treatment of recurrent or metastatic HNSCC
Epstein Barr virus-specific autologous cytotoxic T lymphocytes do not improve survival in nasopharyngeal carcinoma
Related Articles
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com

