https://doi.org/10.55788/04e2f696
Preclinical evidence indicates that BTK inhibition with olverembatinib in addition to venetoclax may be synergistic in acute leukaemia [1]. In a single-arm phase 2 study (NCT05594784), the third-generation BCR-ABL1 tyrosine kinase inhibitor olverembatinib was added to venetoclax and reduced-intensity chemotherapy with vincristine and prednisone as an investigational treatment regimen in 45 participants with newly diagnosed Ph+ ALL. The primary endpoint was the complete molecular response (CMR) rate after 3 x 28-day cycles of therapy [2]. Dr Xiaoyuan Gong (Chinese Academy of Medical Sciences, China) presented the results.
Complete remission (CR; n=43) or CR with incomplete count recovery (CRi; n=2) was reached by all participants. Therefore, all participants were allowed to follow cycles 2 and 3, as per the study protocol. “After 3 cycles of therapy, the CMR rate was 62.2%,” said Dr Gong. In addition, the CMR rates at week 4 and 8 were 53.3% and 60.0%, respectively. Indeed, olverembatinib resulted in quick improvements in haematologic variables during the first therapy cycle (see Figure). Depending on the CMR status of the participant and the availability of stem cell donors, subsequent therapy options were provided. Independent of the subsequent treatment option applied, CMR was achieved in 88.9% of the participants at any time over the course of treatment.
Figure: Rapid improvements in haematologic variables during cycle 1 [2]
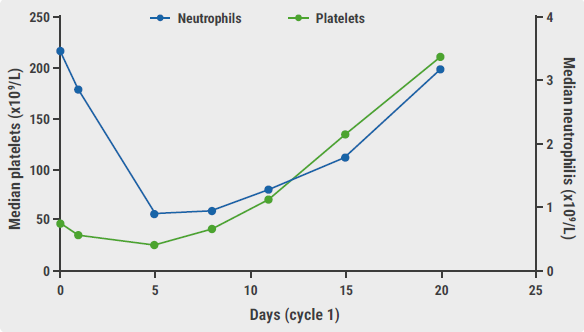
PLT, platelets.
Regarding the safety of olverembatinib combination therapy, grade 3 febrile neutropenia was observed in 22.2% of the participants. The most common adverse events were of grade 1 or 2 and mainly restricted to fatigue and sensory neuropathy. Coronary artery stenosis led to a study discontinuation for 1 participant and 1 participant died due to an infection after autologous stem cell transplant.
Overall, olverembatinib plus venetoclax and reduced chemotherapy appeared safe and effective in previously untreated participants with Ph+ ALL.
- Fang DD, et al. Transl Oncol. 2022;15(1):101244.
- Gong X, et al. Olverembatinib combined with venetoclax and reduced-intensity chemotherapy for patients with newly diagnosed Philadelphia chromosome-positive acute lymphoblastic leukemia: early results from a phase II study. Abstract 827, 65th ASH Annual Meeting, 9–12 December 2023, San Diego, CA, USA.
Copyright ©2024 Medicom Medical Publishers
Posted on
Previous Article
« Undetectable MRD on maintenance venetoclax, acalabrutinib, and obinutuzumab in the majority of R/R CLL participants Next Article
Blinatumomab reduces toxicity in the consolidation phase in paediatric high-risk B-cell ALL »
« Undetectable MRD on maintenance venetoclax, acalabrutinib, and obinutuzumab in the majority of R/R CLL participants Next Article
Blinatumomab reduces toxicity in the consolidation phase in paediatric high-risk B-cell ALL »
Table of Contents: ASH 2023
Featured articles
Meet the Trialist: Prof. Jeff Sharman on ELEVATE-TN
Leukaemia
FLT3-ITD-specific MRD assessment useful for clinical management of AML
MRD status rather than FLT3-ITD co-mutation is linked to the benefit of CR1-allo in NPM1-mutated AML
Promising results for quizartinib, venetoclax, and decitabine in FLT3-ITD mutated AML
AUGMENT-101: Excellent results for revumenib in R/R KMT2Ar leukaemia
Blinatumomab reduces toxicity in the consolidation phase in paediatric high-risk B-cell ALL
Promising results for olverembatinib in combination with venetoclax for Ph+ ALL
Undetectable MRD on maintenance venetoclax, acalabrutinib, and obinutuzumab in the majority of R/R CLL participants
Lymphoma
Is allogeneic stem cell transplantation a solid option in R/R LBCL or R/R T-cell lymphoma?
Encouraging results for the addition of acalabrutinib to lenalidomide and rituximab in follicular lymphoma
Can ibrutinib ameliorate outcomes in R/R ABC-DLBCL undergoing autoSCT?
Primary phase 2 efficacy and safety results of M-Pola in relapsed/refractory LBCL
SYMPATICO: Ibrutinib plus venetoclax boosts PFS in R/R mantle cell lymphoma
Multiple Myeloma
KdD outperforms Kd in R/R MM also in participants with poor renal function
IsKia: Novel treatment regimen for MM delivers high MRD-negativity rates
Novel standard-of-care in newly diagnosed MM
Myeloproliferative Neoplasms
TRANSFORM-1: High spleen volume reduction rates for navitoclax plus ruxolitinib in myelofibrosis
Momelotinib beats controls regarding transfusion outcomes in myelofibrosis
DALIAH: Peginterferon-α head-to-head against hydroxyurea in MPN
Non-Malignant Haematology
Long-term efficacy and safety of iptacopan in PNH with anaemia
ADVANCE IV: Swift responses on efgartigimod in ITP
Favourable QoL and bleeding outcomes for rilzabrutinib in ITP
Novel risk assessment model acts on increasing hospital-acquired venous thromboembolism rates among children
Miscellaneous Topics
Axatilimab may present a new therapeutic strategy in chronic GvHD
Pomalidomide may become the first approved therapy for hereditary haemorrhagic telangiectasia
Ancestry-specific study into CH delivers new leads
Featured Interviews
Interview: Sandwich treatment model shows promise for mantle cell lymphoma
Meet the Trialist: Prof. Jeff Sharman on ELEVATE-TN
Related Articles
June 15, 2023
Long-term success for CD19 CAR T-cell therapy in CLL
February 18, 2021
More complicated course of COVID-19 in leukaemia patients
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com

