https://doi.org/10.55788/5dcb20eb
Lenalidomide plus rituximab is an efficacious first-line therapy in patients with FL [1,2]. “However, it is hypothesised that adding the BTK inhibitor acalabrutinib to lenalidomide and rituximab increases the efficacy due to beneficial effects on the immune microenvironment,” introduced speaker Dr Paolo Strati (MD Anderson Cancer Center, TX, USA).
Dr Strati and co-researchers investigated the safety and efficacy of the combination of the BTK inhibitor acalabrutinib with lenalidomide and rituximab in a phase 2 study (NCT04404088) among 24 participants with previously untreated FL [3]. Enrolled participants received a daily dose of 100 mg acalabrutinib for 13 x 28-day cycles and lenalidomide plus rituximab for 12 cycles. The primary endpoint was a complete response (CR) rate of more than 80%.
The best observed CR rate was 92%, meeting the primary endpoint (see Figure). “We already noticed a CR rate of 62.5% at 3 months, increasing to 92% at 6 months,” added Dr Strati. The 2-year progression-free survival rate was 79% and the 2-year overall survival rate was 92%. “We also observed favourable biological changes in multiple circulating immune cells by adding acalabrutinib to lenalidomide and rituximab,” emphasised Dr Strati.
Figure: Response rates over time, at the end of treatment and timepoint of best response [3]
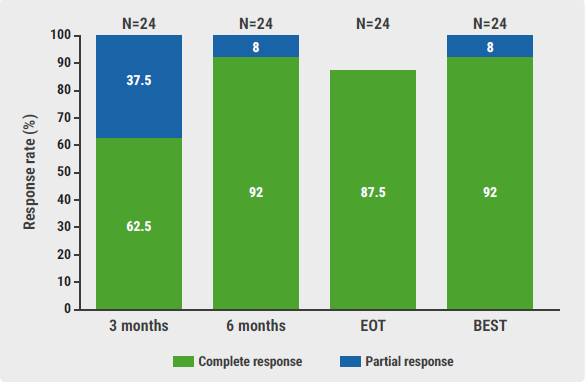
BEST, best response timepoint; CR, complete response; EOT, end of treatment; M, months; PR, partial response.
The most common grade 3–4 treatment-emergent adverse events were neutropenia (58%), ALT elevation (17%), AST elevation (12.5%), and infections (12.5%).
Upon this finding, the study included another 26 participants to further explore this promising treatment option as first-line therapy for participants with FL. The results of this extended study have not been analysed yet.
- Morschhauser F, et al. N Eng J Med 2018;379(10):934-947.
- Strati P, et al. Blood. 2021;137(8):1124-1129.
- Strati P, et al. Addition of acalabrutinib to lenalidomide and rituximab induces high complete response rates in patients with previously untreated follicular lymphoma: results of a phase 2 study. Abstract 983, 65th ASH Annual Meeting, 9–12 December 2023, San Diego, CA, USA.
Copyright ©2024 Medicom Medical Publishers
Posted on
Previous Article
« Can ibrutinib ameliorate outcomes in R/R ABC-DLBCL undergoing autoSCT? Next Article
Is allogeneic stem cell transplantation a solid option in R/R LBCL or R/R T-cell lymphoma? »
« Can ibrutinib ameliorate outcomes in R/R ABC-DLBCL undergoing autoSCT? Next Article
Is allogeneic stem cell transplantation a solid option in R/R LBCL or R/R T-cell lymphoma? »
Table of Contents: ASH 2023
Featured articles
Meet the Trialist: Prof. Jeff Sharman on ELEVATE-TN
Leukaemia
FLT3-ITD-specific MRD assessment useful for clinical management of AML
MRD status rather than FLT3-ITD co-mutation is linked to the benefit of CR1-allo in NPM1-mutated AML
Promising results for quizartinib, venetoclax, and decitabine in FLT3-ITD mutated AML
AUGMENT-101: Excellent results for revumenib in R/R KMT2Ar leukaemia
Blinatumomab reduces toxicity in the consolidation phase in paediatric high-risk B-cell ALL
Promising results for olverembatinib in combination with venetoclax for Ph+ ALL
Undetectable MRD on maintenance venetoclax, acalabrutinib, and obinutuzumab in the majority of R/R CLL participants
Lymphoma
Is allogeneic stem cell transplantation a solid option in R/R LBCL or R/R T-cell lymphoma?
Encouraging results for the addition of acalabrutinib to lenalidomide and rituximab in follicular lymphoma
Can ibrutinib ameliorate outcomes in R/R ABC-DLBCL undergoing autoSCT?
Primary phase 2 efficacy and safety results of M-Pola in relapsed/refractory LBCL
SYMPATICO: Ibrutinib plus venetoclax boosts PFS in R/R mantle cell lymphoma
Multiple Myeloma
KdD outperforms Kd in R/R MM also in participants with poor renal function
IsKia: Novel treatment regimen for MM delivers high MRD-negativity rates
Novel standard-of-care in newly diagnosed MM
Myeloproliferative Neoplasms
TRANSFORM-1: High spleen volume reduction rates for navitoclax plus ruxolitinib in myelofibrosis
Momelotinib beats controls regarding transfusion outcomes in myelofibrosis
DALIAH: Peginterferon-α head-to-head against hydroxyurea in MPN
Non-Malignant Haematology
Long-term efficacy and safety of iptacopan in PNH with anaemia
ADVANCE IV: Swift responses on efgartigimod in ITP
Favourable QoL and bleeding outcomes for rilzabrutinib in ITP
Novel risk assessment model acts on increasing hospital-acquired venous thromboembolism rates among children
Miscellaneous Topics
Axatilimab may present a new therapeutic strategy in chronic GvHD
Pomalidomide may become the first approved therapy for hereditary haemorrhagic telangiectasia
Ancestry-specific study into CH delivers new leads
Featured Interviews
Interview: Sandwich treatment model shows promise for mantle cell lymphoma
Meet the Trialist: Prof. Jeff Sharman on ELEVATE-TN
Related Articles
February 20, 2023
Promising results for triplet therapy with magrolimab in AML

© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com

