The currently recommended first-line ITP treatment is high-dose corticosteroids. However, this therapy is limited by frequent side effects, heterogeneous responses, and high relapse rates. One survey showed that 98% of patients experienced ≥1 side effect and 38% had to stop or reduce dose because of intolerable side effects [2]. Another problem is heterogeneity of responses using corticosteroids, with 30% of patients failing to respond at all. The majority (70–90%) of other patients will relapse at some point, while only 20% of patients remain in sustained long-term remission with this approach. In the United Kingdom, mycophenolate is a commonly used second-line treatment for ITP. Although there is no randomised controlled trial data in ITP, there is retrospective evidence suggesting that mycophenolate is effective in 50-80% of patients and is well tolerated, though responses can be delayed.
In the multicentre, open-label, randomised controlled FLIGHT trial (NCT03156452), 120 patients with ITP (27.5% were >70 years and 15.8% >75 years) requiring first-line treatment were randomised to receive corticosteroids alone (standard care) or combined corticosteroids and mycophenolate. The primary efficacy outcome was time from randomisation to treatment failure, defined as platelets <30 x 109/L and a clinical need for second-line treatment (included refractory and relapsed ITP).
Significantly fewer treatment failures occurred in patients randomised to mycophenolate (22% vs 44%; adjusted HR 0.41; P=0.0064; see Figure). With mycophenolate, significantly more patients responded and less were refractory. “Interestingly, 2 weeks after randomisation, the responses were very similar in both groups,” Dr Charlotte Bradbury (University of Bristol, United Kingdom) added. “This very much reflects the slower mechanism of action of mycophenolate.”
Figure: Proportion of patients without treatment failure in FLIGHT [1]
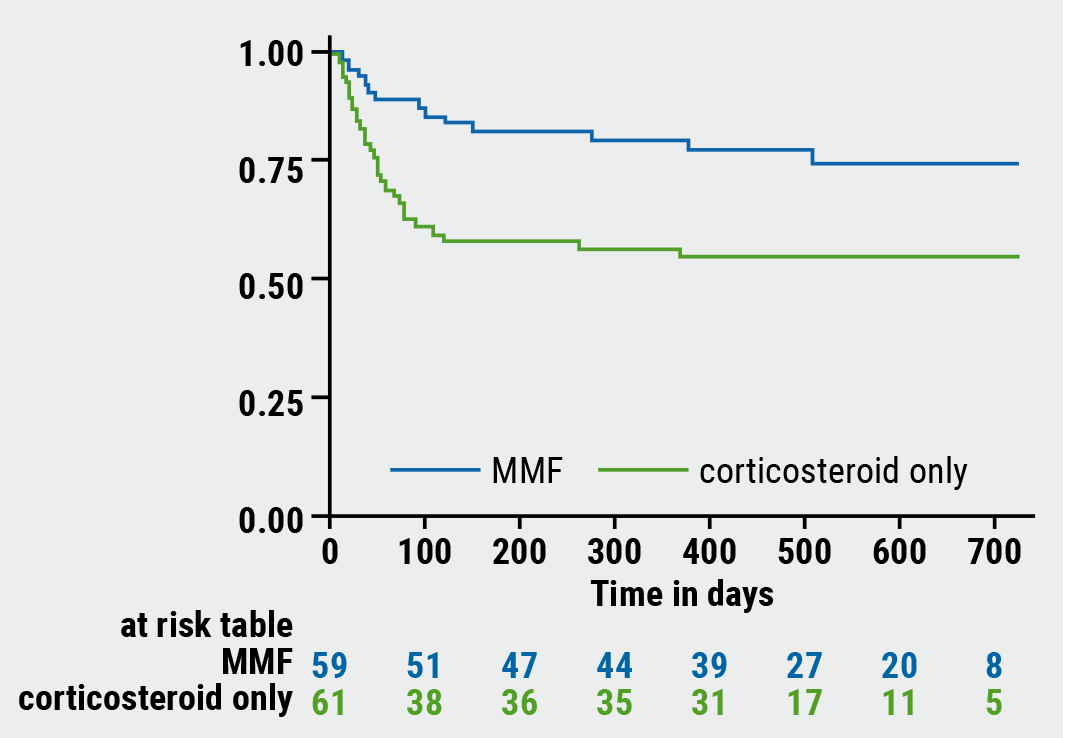
MMF, mycophenolate
Similar rates were observed between the groups regarding treatment side effects and bleeding events. There were no intracranial haemorrhages or fatal bleeds. Of interest, infection occurred in 14 patients in each group, whereas neutropenia occurred only in the corticosteroid-only group (n=4). No patients underwent a splenectomy during follow-up.
Patient-reported outcomes on bleeding (FACT Th6) and quality of life (ICECAP) were quite similar in both groups. Some aspects of quality of life seemed to be worse in patients assigned to the mycophenolate group, including physical role (SF36, P=0.012), mental function (SF36, P=0.519), and fatigue (FACIT-F, P=0.05).
At the final follow-up, 56% of patients treated with corticosteroids alone had not required second-line treatment, which is higher than previous reports. “So, the corticosteroid alone group did really good in this trial,” Dr Bradbury concluded. “It is unclear why some aspects of quality of life seemed to be worse in the mycophenolate group. However, it is an important reminder that disease response and patient experience may not correlate. It also emphasises the importance of including patient-reported outcomes measures within trials.”
- Bradbury CA, et al. A Multicentre Randomised Trial of First Line Treatment Pathways for Newly Diagnosed Immune Thrombocytopenia: Standard Steroid Treatment Versus Combined Steroid and Mycophenolate. the Flight Trial. 62nd ASH Annual Meeting, 5-8 December 2020. Abstract LBA-2.
- Brown TM, et al. BMC Blood Disord. 2012;12:2.
Posted on
Previous Article
« First-in-class antibody sutimlimab selectively inhibits classical complement pathway Next Article
New insights into genetics of MPN »
« First-in-class antibody sutimlimab selectively inhibits classical complement pathway Next Article
New insights into genetics of MPN »
Table of Contents: ASH 2020
Featured articles
COVID-19
More complicated course of COVID-19 in leukaemia patients
Older age and imatinib treatment associated with COVID-19 mortality in CML
Allogeneic SARS-CoV-2-specific T cells to treat COVID-19
More severe COVID-19 outcomes for patients with haematologic malignancies
Acute Lymphoblastic Leukaemia
Improved outcomes, but still substantial part experiences relapses
Strong correlation between peripheral blood and bone marrow NGS MRD
Encouraging outcomes after autoHCT in patients with ALL
Acute Myeloid Leukaemia
Prognostic validity of AML composite model in predicting mortality
Venetoclax plus hypomethylating agents in favourable-risk AML
Encouraging clinical activity of decitabine plus ipilimumab in R/R or secondary MDS/AML
AML patients with specific mutations are unlikely to achieve MRD
Comparable outcomes with gilteritinib or quizartinib in R/R AML
First-in-class macrophage immune checkpoint inhibitor in AML
Bispecific DART® as salvage therapy for primary induction failure and early relapse
Gilteritinib in R/R AML patients priorly treated with midostaurin or sorafenib
Addition of venetoclax provides an effective, lower-intensity regimen
Chronic Leukaemia
Bosutinib effective and well tolerated in newly diagnosed CP-CML
Efficacy and safety of ponatinib in patients with CP-CML who failed second-generation TKIs
First-in-class STAMP inhibitor versus bosutinib in resistant or intolerant CML
PFS and ORR benefits of first-line ibrutinib-based treatment in CLL
Multiple Myeloma
Validation of MY-RADS response assessment category criteria
High symptom burden in transplant-ineligible patients with newly diagnosed MM
Added value of ixazomib to lenalidomide plus dexamethasone in transplant-ineligible newly diagnosed MM
Survival of transplant-eligible newly diagnosed MM in FORTE trial
Better survival with upfront autoSCT versus bortezomib-based intensification
Subcutaneous daratumumab plus pomalidomide and dexamethasone in R/R MM
Melflufen well tolerated with encouraging activity in heavily pretreated R/R MM
Initial data of FcRH5/CD3 T-cell-engaging bispecific antibody
Lymphoma
CD58 aberrations limit durable responses to CD19 CAR T-cell therapy
Anti-CD19 CAR T-cell therapy in relapsed/refractory indolent NHL
Myeloproliferative Neoplasms
MPN disease burden, quality of life, and treatment patterns
Interventions in JAK/STAT signalling pathway
Novel, orally available inhibitor of BCL-XL/BCL-2
New insights into genetics of MPN
Immune Thrombocytopenia
Mycophenolate efficacious and tolerable, even in elderly patients
First-in-class antibody sutimlimab selectively inhibits classical complement pathway
BTK inhibition provides clinically active and durable platelet response
Haemophilia, Sickle Cell Disease, Thalassaemia
First results from gene therapy trial in haemophilia B
Impact of haemophilia on children and their caregivers
Promising CRISPR gene editing results in β-thalassaemia and sickle cell disease
Erythroid maturation agent in patients with β-thalassaemia requiring regular RBC transfusions
Related Articles
February 18, 2021
New insights into genetics of MPN
February 18, 2021
Allogeneic SARS-CoV-2-specific T cells to treat COVID-19

February 18, 2021
Validation of MY-RADS response assessment category criteria
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com

