Dr Lissy de Ridder (Erasmus University Medical Center, Rotterdam, the Netherlands) presented the practice-changing results of the multicentre open-label randomised controlled TISKids study [1]. The study aimed to acquire early control of the inflammatory cascade by inducing early mucosal healing an preventing the accumulation of bowel damage characteristic of the disease. The investigators’ hypothesis was that initiation of infliximab directly after diagnosis in moderate-to-severe paediatric CD patients would prevent the inflammatory cascade, and result in disease control.
Children (n=97) between the ages of 3-17 years with untreated moderate-to-severe CD (defined as a weighted paediatric CD activity index (wPCDAI) >40) were randomised to one of 2 arms: the top-down arm treated with infliximab biosimilar CT-P13 vs the step-up treatment arm. The top-down group (n=50) received 3 infusions of infliximab in the first 6 weeks and 2 maintenance doses at weeks 14 and 22 while being supported by azathioprine for the entire 52 weeks. Patients in the step-up arm started with either prednisone (n=19) or exclusive enteric nutrition (n=28), in addition to azathioprine. Endoscopy was performed at 10 weeks (required) and voluntarily at 52 weeks. Treatment intensification was allowed under certain conditions. Primary non-response was defined as no response at week 6 compared with baseline with the decrease in wPCDAI of <17.5. A secondary loss of response was defined as either an increase of wPCDAI of >17.5 or a total wPCDAI score of >40 after response had already been achieved. The treatment was intensified with (re)start of corticosteroids, (re)start of biological treatment, or intensification of infliximab scheme with a dose intensification or treatment adjustment. The primary endpoint was clinical remission with a wPCDAI score <12.5 at week 52 without the need for additional CD-related therapy or surgery. Secondary endpoints were mucosal healing rates at week 10, as measured by calprotectin levels, and confirmed by endoscopy, as well as the cumulative therapy use (e.g. steroids, biologicals).
Figure. Patients in clinical remission (wPCDAI score <12.5)
without treatment intensification at week 52 [1]
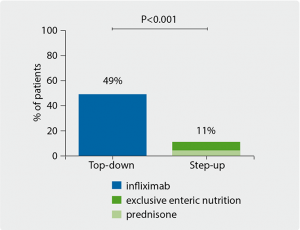
Endoscopic imaging at week 10 indicated that infliximab provided a significant clinical benefit in mucosal healing; 61% of remission the patients in the top-down group had endoscopic remission vs only 14% of the step-up group (P=0.001). The top-down group also had significantly decreased levels of inflammatory markers such as CRP (P=0.008), ESR (P=0.001), leukocyte counts (P<0.001), and calprotectin levels (P=0.001). At week 52 the primary endpoint was met, with 49% of the top-down group in full clinical remission, as opposed to 11% of the step-up group (P<0.001) who did not require additional treatment or surgery (see Figure). The patients in the step-up arm also required earlier and more frequent interventions with treatment intensification (P=0.001).
Dr de Ridder pointed out that all patients will be followed up for 5 years, and the true value of early intervention in modifying the inflammatory cascade will only be clear with longer term results. In addition, detailed immunological profiling of all patients will hopefully help stratify patients into those that may benefit from a given treatment; the fact that 11% of the step-up group did achieve remission at 52 weeks suggests that there may be a risk for overtreatment in certain individuals.
- De Ridder L et al. UEG Week 2019, Abstract OP001.
Posted on
Previous Article
« Ustekinumab is safe and effective in ulcerative colitis: 2-year data Next Article
Novel targets in mesothelioma may aid in developing treatment strategies »
« Ustekinumab is safe and effective in ulcerative colitis: 2-year data Next Article
Novel targets in mesothelioma may aid in developing treatment strategies »
Table of Contents: UEGW 2019
Featured articles
Interview with UEG President Prof. Paul Fockens
Upper GI Disorders
Locally active corticosteroid promising in eosinophilic oesophagitis
First-in-human radiofrequency vapor ablation in Barrett’s oesophagus
Irritable Bowel Syndrome
Faecal microbiota transplantation is effective for irritable bowel syndrome
Human milk oligosaccharides improve IBS symptoms
Inflammatory Bowel Disease
Ustekinumab is safe and effective in ulcerative colitis: 2-year data
Decreased microvilli length in CD patients
Phase 2 data shows benefit for mirikizumab in CD patients
Subcutaneous ustekinumab as maintenance therapy in UC
First evidence of long-term efficacy of ABX464 in ulcerative colitis
New treatment may reverse coeliac disease
IBD prevalence 3 times higher than estimated and expected to rise
Microbiome and Microbiota
Early stages of gastric metaplasia: molecular profiling
Plant-based foods and Mediterranean diet associated with healthy gut microbiome
Antibiotic resistance in H. pylori has doubled over last 20 years
Pancreatitis
New model predicts recurrence of acute biliary pancreatitis
Hepatology
Restrictive strategy for cholecystectomy selection does not reduce pain, but does reduce surgery
β-blockers may halt cirrhosis progression: PREDESCI trial
Obeticholic acid prevents liver fibrosis from NASH
Oncology
Metal stents are better than plastic for endoscopic biliary drainage
Ramosetron relieves low anterior resection syndrome
Immunonutrition during neoadjuvant oesophagogastric cancer therapy: no benefit
Endoscopy
EUS-guided histological specimens from the pancreatic cyst wall
Digital single-operator cholangioscopy more sensitive than endoscopic retrograde cholangiopancreatography
New single-use duodenoscope well-liked by endoscopists
Related Articles
October 23, 2019
Phase 2 data shows benefit for mirikizumab in CD patients
October 23, 2019
β-blockers may halt cirrhosis progression: PREDESCI trial
October 23, 2019
First-in-human radiofrequency vapor ablation in Barrett’s oesophagus
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com

