"These findings are an important contribution to the field, showing a safe strategy to incorporate venetoclax into the most common induction regimen used to treat newly diagnosed acute myeloid leukemia internationally," researchers write in The Lancet Haematology.
The phase-2 study included 33 patients (median age, 40 years) with previously untreated de novo AML and an ECOG performance status of 0 to 2.
They received induction treatment with intravenous daunorubicin (60 mg/m2 on days 1 to 3), intravenous cytarabine (100 mg/m2 on days 1 to 7), and oral venetoclax (100 mg on day 4, 200 mg on day 5, and 400 mg on days 6 to 11), the so-called "DAV" regimen.
Overall, the complete remission rate after one DAV treatment cycle was 91%, report Dr. Hong-Hu Zhu of Zhejiang University School of Medicine, in Hangzhou, and colleagues.
Complete remission rates were 100%, 83% and 75% in European LeukemiaNet (ELN) favorable-, intermediate-, and poor-risk groups, respectively.
Of the 30 patients who achieved complete remission, 29 (97%) had undetectable measurable residual disease.
In addition, patients showed "rapid recovery" from myelosuppression, with a median time to recovery of 21 days following induction therapy.
All patients developed neutropenia, thrombocytopenia and anemia. Other adverse events included febrile neutropenia (55%), pneumonia (21%) and sepsis (12%). No treatment-related deaths occurred.
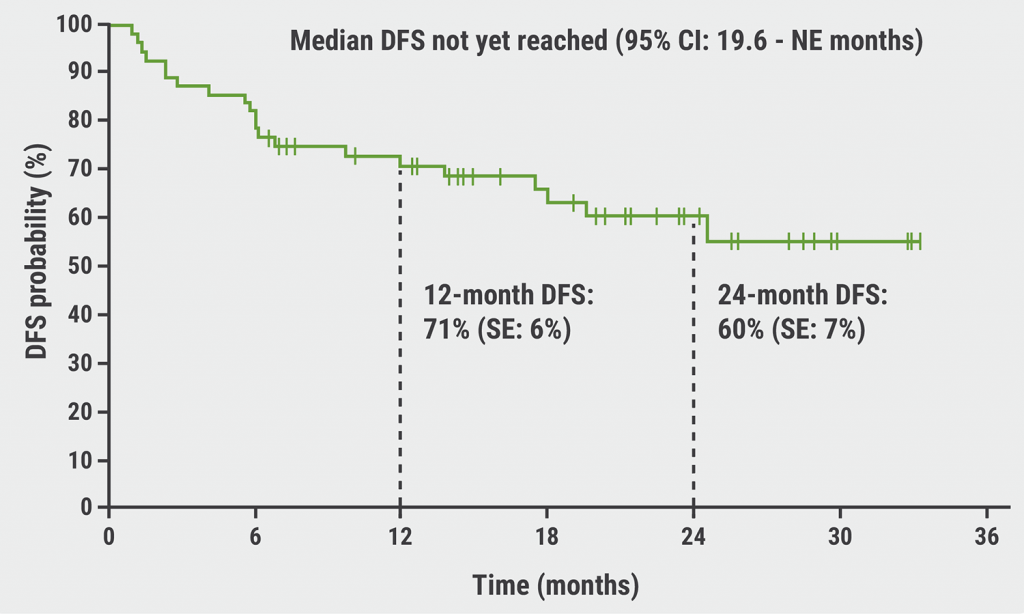
With a median follow-up of 11 months, estimated one-year overall survival was 97% and one-year event-free survival was 72%. The researchers caution that the follow-up time was too short to adequately assess survival benefit.
Based on the "encouraging" results from this phase-2 study, they have initiated a randomized controlled trial to compare the DAV and DA regimens in newly diagnosed adults with AML.
The co-authors of an accompanying comment note that induction chemotherapy with an anthracycline and cytarabine have been the standard of care for AML for more than five decades.
In the past five years, venetoclax, an inhibitor of the anti-apoptotic B-cell lymphoma-2 (BCL-2) protein, has "revolutionized" the management of AML, say Dr. Kieran Sahasrabudhe and Dr. Alice Mims with The Ohio State University Comprehensive Cancer Center in Columbus.
The new study shows "favorable response rates, a high rate of measurable residual disease-negativity among patients with response, favorable survival data, and manageable adverse effects" with the DAV regimen.
"Larger studies with longer follow-up are needed to further validate this regimen, including the planned randomized trial comparing daunorubicin, cytarabine, and venetoclax with daunorubicin and cytarabine alone," Drs. Sahasrabudhe and Mims say.
"Although several additional questions remain regarding the optimization of this treatment approach, the role of venetoclax in the management of AML is likely to continue to expand as additional investigation provides further insights into the therapeutic potential of this agent," they add.
The study was funded by the Leading Innovative and Entrepreneur Team Introduction Program of Zhejiang and the National Natural Science Foundation of China. The researchers did not disclose any significant financial relationships.
SOURCE: https://bit.ly/3Pdas5g and https://bit.ly/3vXFp5E The Lancet Haematology, online May 2, 2022.
By Reuters Staff
Posted on
Previous Article
« CAR-T outcomes similar among Black, Hispanic, white, Asian patients Next Article
E-cigarettes do not appear to confer heart benefits to users who also smoke traditional cigarettes »
« CAR-T outcomes similar among Black, Hispanic, white, Asian patients Next Article
E-cigarettes do not appear to confer heart benefits to users who also smoke traditional cigarettes »
Related Articles
August 28, 2020
Venetoclax + navitoclax promising for R/R ALL or LL
February 18, 2021
PFS and ORR benefits of first-line ibrutinib-based treatment in CLL
February 4, 2022
Encouraging results of novel triplet combination for AML
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy

