https://doi.org/10.55788/7a1e3928
Prof. Justin Ezekowitz (University of Alberta, Canada) presented the results of the SODIUM-HF (NCT02012179) trial, which was simultaneously published in The Lancet [1,2]. Participants were randomly assigned (1:1) to either usual care according to local guidelines (n=409) or a low-sodium diet <1,500 mg/day for 1 year (n=397). The mean age of the participants was 66 years, 32% were women and 35% had diabetes. The primary endpoint was the composite of cardiovascular-related admission to hospital, cardiovascular-related emergency department visit, or all-cause death within 12 months. Enrollment was stopped early after only about half of the planned patient population had been recruited due to the COVID-19 pandemic.
At 12 months, primary outcome events had occurred in 15% in the low-sodium group, as compared with 17% in the control group (HR 0.89; 95% CI 0.63–1.26; P=0.53). Likewise, death from any cause was not different between the groups (6% of participants in the low-sodium group vs 4% in the usual care group; HR 1.38; 95% CI 0.73–2.60; P=0.32). Also, no significant difference were seen in cardiovascular-related hospitalisation (10% low-sodium group vs 12% control group; HR 0.82; 95% CI 0.54–1.24; P=0.36) and cardiovascular-related emergency department visits (4% in both groups; HR 1.21; 95% CI 0.60–2.41; P=0.60). Subgroup analyses did not reveal any meaningful differences. There were no safety events in either group. However, as a QoL measure, the Kansas City Cardiomyopathy Questionnaire (KCCQ) revealed a difference of 3.38 points from baseline to 12 months in the overall score, 3.29 points in the clinical summary score, and 3.77 points in the physical limitation score, indicating an overall QoL improvement for the low-sodium group.
In summary, a dietary intervention to reduce sodium intake did not reduce HF-related clinical events in this trial. Prof. Ezekowitz concluded that while the study was underpowered given a low number of events to detect a meaningful difference statistically, observed differences in QoL warrant further investigation.
- Ezekowitz JA, et al. SODIUM-HF: Study Of Dietary Intervention Under 100 Mmol In Heart Failure. Abstract 402–11, ACC 2022, 2–4 April, Washington DC, USA.
- Ezekowitz JA, et al. Lancet 2022;399(10333):1391–1400.
Copyright ©2022 Medicom Medical Publishers
Posted on
Previous Article
« Sotagliflozin SCORED MACE benefit Next Article
Replacing septal reduction therapy with mavacamten for HCM »
« Sotagliflozin SCORED MACE benefit Next Article
Replacing septal reduction therapy with mavacamten for HCM »
Table of Contents: ACC 2022
Featured articles
Alirocumab significantly reduces high-risk coronary plaques
Highlighted Original Research
POISE-3: Tranexamic acid for non-cardiac surgery
Treating chronic mild hypertension during pregnancy leads to better outcomes
New VOYAGER PAD data: Should patients with both PAD and CKD get rivaroxaban?
Alirocumab significantly reduces high-risk coronary plaques
Aggressive warming during non-cardiac surgery does not improve outcomes
Heart Failure and Cardiomyopathy
DIAMOND trial: Patiromer lowers risk of severe hyperkalaemia
Replacing septal reduction therapy with mavacamten for HCM
Omecamtiv mecarbil does not impact exercise capacity of patients with HFrEF
Symptomatic obstructive hypertrophic cardiomyopathy: long-term mavacamten control
Interventional and Structural Cardiology
COMPLETE revascularisation improves angina-related QoL
Plot twist for negative FAME 3 results: early QoL benefits of PCI
1-year CLASP TR results support tricuspid regurgitation repair
Head-to-head: post-TAVR edoxaban not better than DAPT
Chocolate Touch vs Lutonix catheters
No FLAVOUR difference between FFR and IVUS for PCI guidance
Myocardial Infarction
Low-resource countries benefit from global STEMI initiative
Sodium thiosulfate ineffective at cardiac protection
ICM-guided management did not improve MACE after MI
Prevention
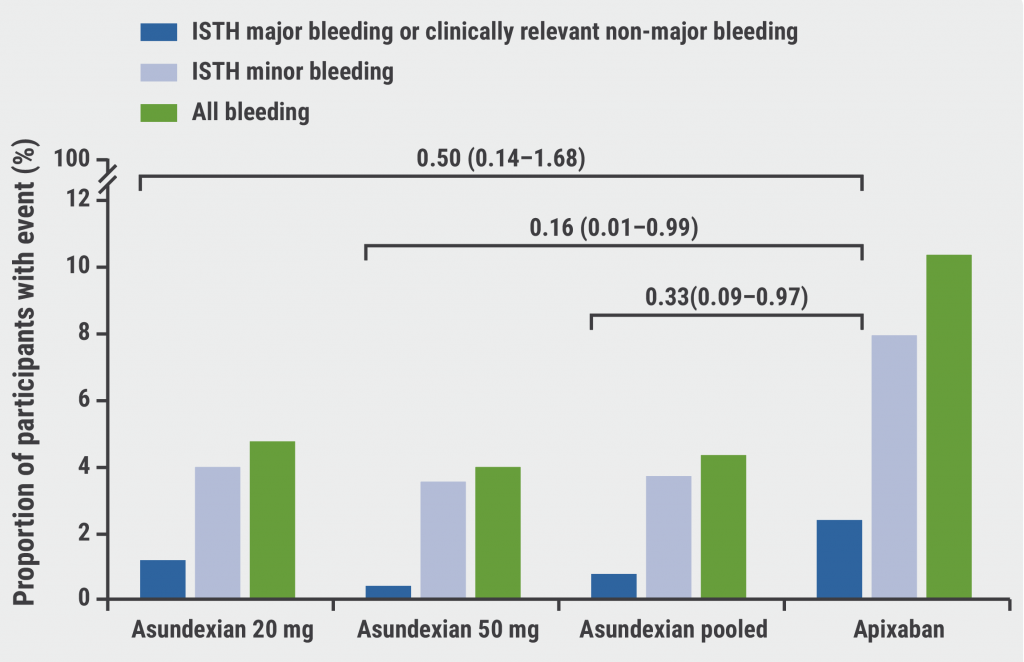
PACIFIC-AF: Low bleeding rates for asundexian in atrial fibrillation
RCT-IVVE trial: Do HF patients benefit from annual flu shots?
TRANSLATE-TIMI 70: Primary endpoint met but safety concerns for vupanorsen
Lipoprotein(a) slashed by 98% in APOLLO trial
Dietary intervention from your supermarket
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com