https://doi.org/10.55788/0fb6234c
“Most patients with HFpEF are overweight or have obesity,” stated Dr Mikhail Kosiborod (University of Missouri, MO, USA) [1]. “Since there are no approved therapies for the obesity phenotype of HFpEF and it has been demonstrated that the GLP-1 agonist semaglutide induces weight loss in overweight and obese individuals, the STEP-HFpEF trial was conducted” [1–3]. This phase 3 trial (NCT04788511) investigated the effects of 2.4 mg semaglutide, subcutaneously administered once weekly, on HFpEF symptoms and physical functioning in patients with the obesity HFpEF phenotype [3]. The double-blind trial randomised 529 adult patients 1:1 to the experimental arm or a placebo. The co-primary endpoints were the change in Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ-CSS) and the percentage change in body weight from baseline to week 52.
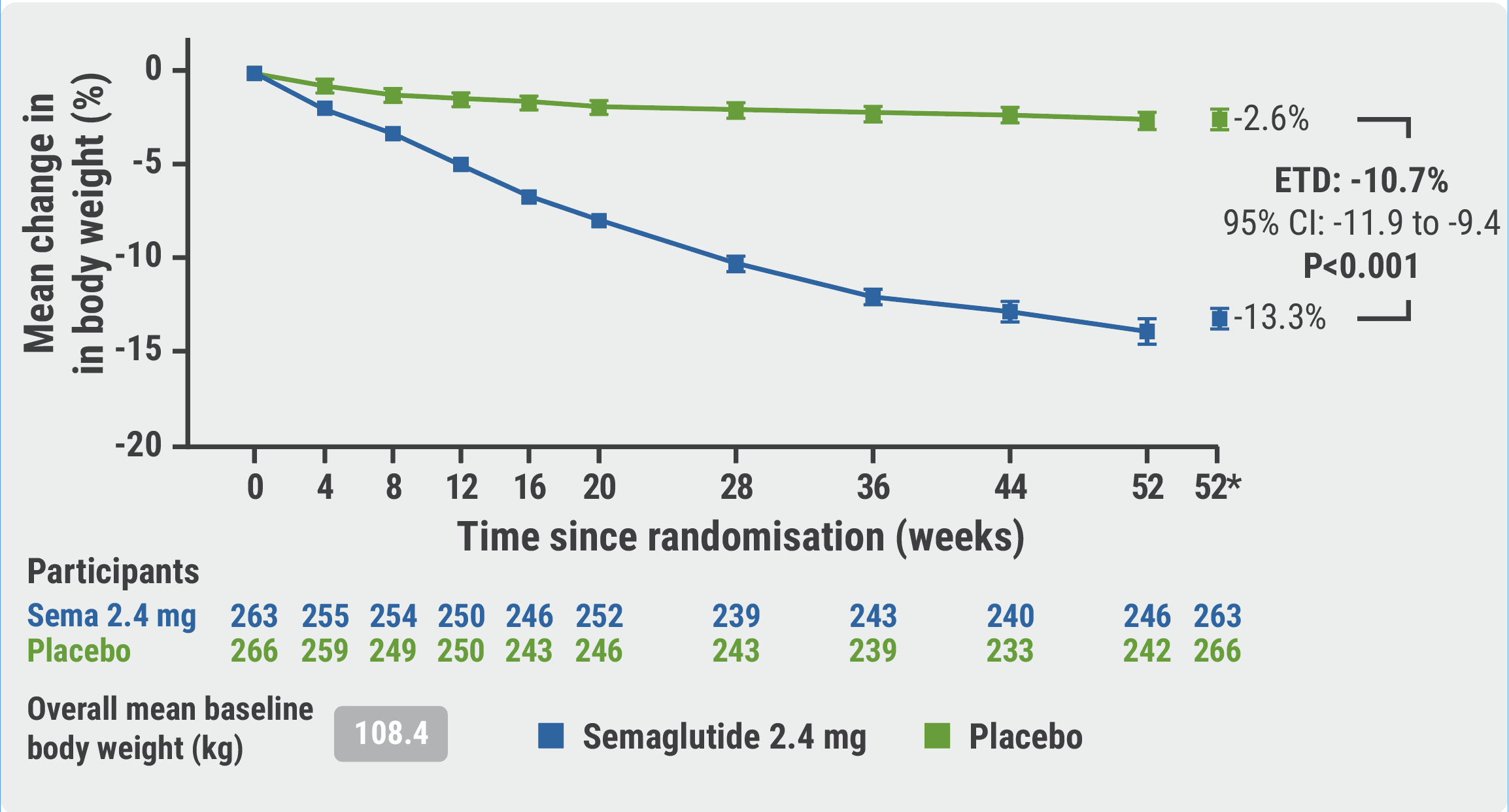
The mean change in KCCQ-CSS at week 52 was 16.6 points in the active treatment arm and 8.7 points in the placebo arm, representing a significant 7.8-point benefit for the semaglutide arm (95% CI 4.8–10.9; P<0.001; see Figure 1). Further, body weight was reduced by 13.3% in the semaglutide arm compared with 2.6% in the placebo arm, meeting this second primary endpoint (95% CI -11.9 to -9.4; P<0.001; see Figure 2). Participants receiving semaglutide also showed a significant improvement in 6-minute walking distance compared with those receiving placebo (mean difference 20.3 metres; 95% CI 8.6–32.1; P<0.001). At week 52, CRP levels had dropped to a ratio of 0.93 compared withbaseline in the placebo arm and to a ratio of 0.56 compared with baseline in the semaglutide arm. Mean reductions from baseline NT-proBNP were approximately 5% and 21% in the placebo and semaglutide arms, respectively.
Figure 1: Change in KCCQ-CSS from baseline to week 52 [1]

ETD, estimated treatment difference; KCCQ-CSS, Kansas City Cardiomyopathy Questionnaire Clinical Summary Score.
Figure 2: Change in body weight from baseline to week 52 [1]
ETD, estimated treatment difference.
Serious adverse events (AEs) were more common in the placebo arm than in the semaglutide arm (26.7% vs 13.3%; P<0.001). This effect was mainly driven by a higher rate of cardiac disorders in the placebo arm (11.3% vs 2.7%; P<0.001). The AE rate of participants discontinuing treatment appeared to be higher in the experimental arm (13.3% vs 5.3%), mainly due to a difference in gastrointestinal AEs resulting in treatment discontinuations (9.5% vs 2.6%).
Discussant of the trial, Prof. Frank Ruschitzka (University Heart Center Zürich, Switzerland) congratulated the investigators on these potentially practice-changing results but wondered whether the effects of semaglutide on HFpEF symptoms are mainly driven by weight loss or by other mechanisms. He also warned that weight loss is not a great proxy for fat loss and that the loss of lean mass and the risk for sarcopaenia need to be further investigated. Finally, he stressed that the proportion of non-White individuals in the STEP-HFpEF trial was low and that the effects of semaglutide on the HFpEF with obesity phenotype need to be addressed in other ethnicities.
- Kosiborod MN, et al. Semaglutide in patients with heart failure with preserved ejection fraction and obesity. Hot Line Session 1, ESC Congress 2023, 25–28 August, Amsterdam, the Netherlands.
- Dunlay SM, et al. Nat Rev Cardiol. 2017;14(10):591–602.
- Kushner RF, et al. Obesity (Silver Spring). 2020;28:1050–1061.
Copyright ©2023 Medicom Medical Publishers
Posted on
Previous Article
« Guidelines for the management of endocarditis Next Article
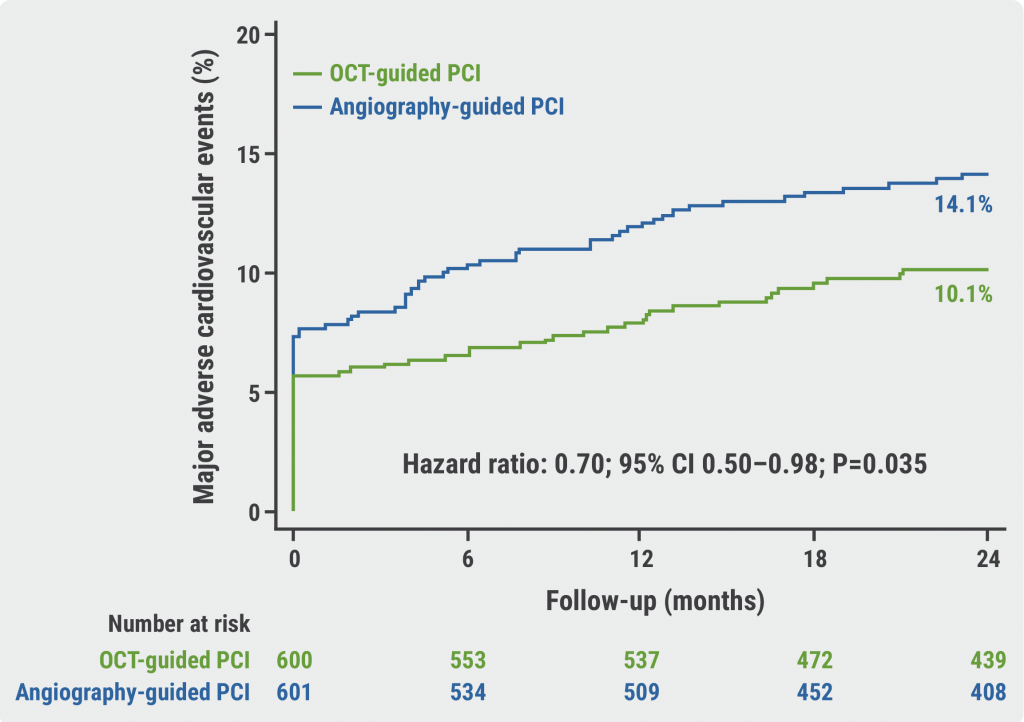
OCTOBER trial: OCT-guided PCI improves clinical outcomes in bifurcation lesions »
« Guidelines for the management of endocarditis Next Article
OCTOBER trial: OCT-guided PCI improves clinical outcomes in bifurcation lesions »
Table of Contents: ESC 2023
Featured articles
How to manage arterial thrombosis and thromboembolism in COVID-19?
2023 ESC Guidelines & Updates
Heart failure: the 2023 update
Guidelines for Acute Coronary Syndrome
Guidelines for the management of cardiomyopathies
Cardiovascular disease and diabetes: new guidelines
Guidelines for the management of endocarditis
Trial Updates in Heart Failure
Traditional Chinese medicine successful in HFrEF
CRT upgrade benefits patients with HFrEF and an ICD
Catheter ablation saves lives in end-stage HF with AF
Meta-analysis: Does FCM improve clinical outcomes in HF?
HEART-FID: Is intravenous ferric carboxymaltose helpful in HFrEF with iron deficiency?
Natriuresis-guided diuretic therapy to facilitate decongestion in acute HF
DICTATE-AHF: Early dapagliflozin to manage acute HF
STEP-HFpEF: Semaglutide safe and efficacious in HFpEF plus obesity
Key Research on Prevention
Does colchicine prevent perioperative AF and MINS?
Diagnostic tool doubles cardiovascular diagnoses in patients with COPD or diabetes
Inorganic nitrate strongly reduces CIN in high-risk patients undergoing angiography
Finetuning Antiplatelet and Anticoagulation Therapy
Should we use anticoagulation in AHRE to prevent stroke?
Results of FRAIL-AF trial suggest increased bleeding risk with DOACs
The optimal duration of anticoagulation therapy in cancer patients with DVT
DAPT or clopidogrel monotherapy after stenting in high-risk East-Asian patients?
Assets for ACS and PCI Optimisation
Immediate or staged revascularisation in STEMI plus multivessel disease?
Lp(a) and cardiovascular events: which test is the best?
No benefit of extracorporeal life support in MI plus cardiogenic shock
Functional revascularisation outperforms culprit-only strategy in older MI patients
Can aspirin be omitted after PCI in patients with high bleeding risk?
Angiography vs OCT vs IVUS guidance for PCI: a network meta-analysis
OCTOBER trial: OCT-guided PCI improves clinical outcomes in bifurcation lesions
Other
Minimising atrial pacing does not reduce the risk for AF in sinus node disease
ARAMIS: Can anakinra alleviate acute myocarditis?
Expedited transfer to a specialised centre does not improve cardiac arrest outcomes
Acoramidis improves survival and functional status in ATTR-CM
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com