The study, involving 1,405 adults, demonstrated that the drug, zavegepant, was superior to placebo in relieving pain and most other "bothersome" symptoms including nausea or sensitivity to light or sound, the company said.
A single intranasal dose of the drug helped relieve pain as early as 15 minutes, lasting for 48 hours after the treatment, Biohaven said.
Around one billion people worldwide suffer from migraine, according to the Migraine Research Foundation.
Biohaven's Nurtec ODT, has already been approved for the treatment and prevention of migraine in the United States. Other drugs for treatment include AbbVie's Ubrelvy and Impel NeuroPharma's recently approved intranasal spray, Trudhesa.
Pfizer Inc would acquire overseas marketing rights to zavegepant and rimegepant, branded as Nurtec ODT, from Biohaven, the companies said last month.
Biohaven said it plans to file a marketing application for zavegepant with the U.S. Food and Drug Administration in the first quarter of next year.
By Reuters Staff
Posted on
Previous Article
« Is recovery of sexual function after radical prostatectomy surgeon dependent? Next Article
High-frequency spinal cord stimulation provides lasting relief of painful diabetic neuropathy »
« Is recovery of sexual function after radical prostatectomy surgeon dependent? Next Article
High-frequency spinal cord stimulation provides lasting relief of painful diabetic neuropathy »
Related Articles
September 10, 2020
Migraine as a cyclical functional disorder
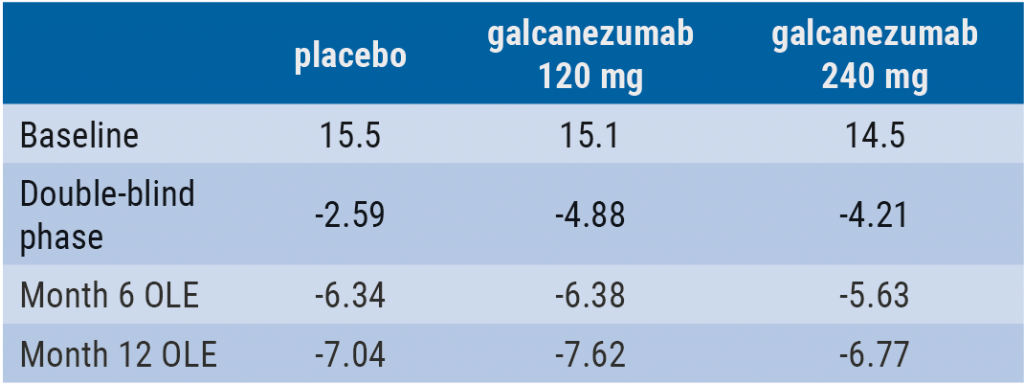
August 27, 2019
Galcanezumab reduces healthcare resource utilisation
January 5, 2023
Atogepant for the preventive treatment of chronic migraine
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy

