https://doi.org/10.55788/490b7ec3
The investigational agent ELA026 targets signal regulatory protein (SIRP)-α/β1 on myeloid cells and SIRPγ on T-lymphocytes. Dr Abhishek Maiti (MD Anderson Cancer Center, TX, USA) and colleagues tested ELA026 among patients with secondary HLH in a phase 1 study (NCT05416307) [1]. During the current presentation, Dr Maiti discussed findings from 8 treatment-naïve participants with malignancy-associated HLH exposed to ELA026. “These participants stemmed from the refined study population cohort,” said Dr Maiti. For the safety analysis, data from cohorts 1 and 2 was involved, including participants with refractory or relapsed disease.
After 4 weeks of therapy, all 8 participants presented with at least a partial response. The observed 2-month survival rate of 88% on ELA026 was substantially higher than the 2-month survival rate from natural history malignancy-associated HLH cohorts (50%; see Figure).
Figure: Therapy response and survival upon ELA026 administration to participants with treatment-naïve malignancy-associated HLH [1]
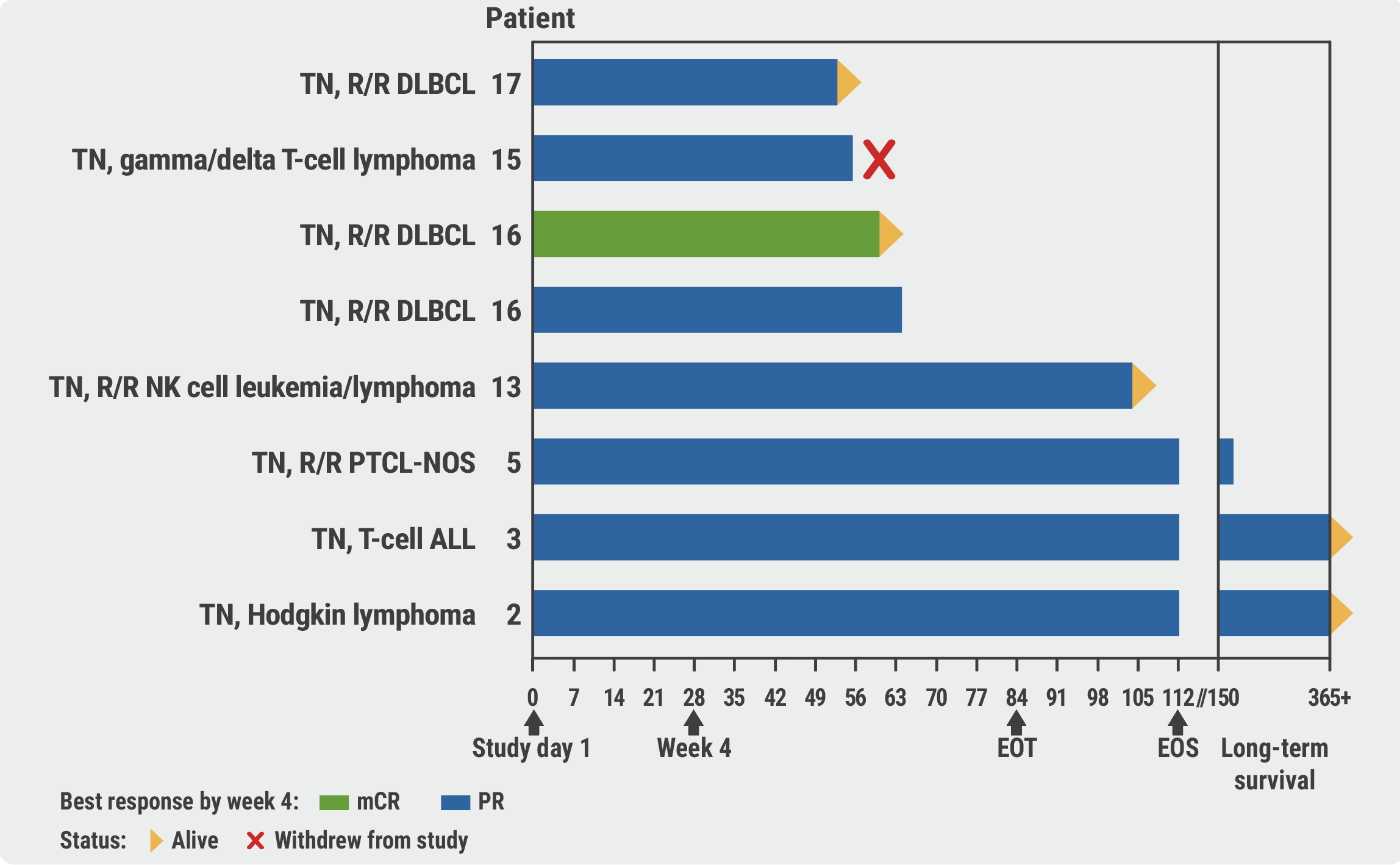
AITL, angioimmunoblastic T-cell lymphoma; ALL, acute lymphoblastic leukaemia; CR, complete remission; DLBCL, diffuse large B-cell lymphoma; EOS, end of study; EOT, end of treatment; HI, haematologic improvement; mCR, marrow complete remission; NK cell, natural killer cell; PR, partial remission; PTCL-NOS, peripheral T-cell lymphoma not otherwise specified; R/R, relapsed/refractory; TN, treatment-naïve.
Thrombocytopenia (35%), neutropenia (24%), and infusion-related reactions (18%) were among the most commonly observed adverse events (AEs) in the 17 participants included in the safety analysis. “A third of the neutropenia and thrombocytopenia was already established at baseline,” highlighted Dr Maiti.
“ELA026 appeared well-tolerated and was associated with improved survival outcomes among treatment-naïve participants with malignancy-associated HLH,” concluded Dr Maiti. “This promising therapy may help to control the cytokine storm from any trigger that is the main driver of early mortality in patients with secondary HLH.”
- Maiti A, et al. ELA-026 targeting of SIRP+ immune cells results in a high response rate and improved 2-month survival of treatment-naïve malignancy associated hemophagocytic lymhohistiocytosis in a phase 1 study. LB3442, EHA congress 2024, 13–16 June, Madrid, Spain.
Copyright ©2024 Medicom Medical Publishers
Posted on
Previous Article
« PhALLCON: Third-generation TKI superior to first-generation TKI in Ph+ ALL Next Article
RUBY: Promising data for first AsCas12a gene-editing therapy in sickle cell disease »
« PhALLCON: Third-generation TKI superior to first-generation TKI in Ph+ ALL Next Article
RUBY: Promising data for first AsCas12a gene-editing therapy in sickle cell disease »
Table of Contents: EHA 2024
Featured articles
Meet the Expert: Prof. C. Ola Landgren discusses MRD as a key endpoint in haematological cancer trials
Multiple Myeloma
Isa-VRd proves its value in newly diagnosed MM in the IMROZ trial
PERSEUS: High MRD negativity rates with D-VRd and consolidation therapy and D-R maintenance in MM
Post-intensification data confirm superiority of quadruple therapy in MM
Promising phase 1 results for novel CAR T-cell therapy in MM
DREAMM 8: Belantamab mafodotin offers hope for patients with RRMM
Leukaemia
PhALLCON: Third-generation TKI superior to first-generation TKI in Ph+ ALL
APOLLO: ATRA plus ATO meets expectations in high-risk APL
Excellent phase 3 results for asciminib in chronic myeloid leukaemia
AUGMENT-101: Revumenib trial in KMT2Ar leukaemia stopped early for efficacy
FLAG-Ida plus venetoclax induces high MRD-negativity rates in AML
CD40/CD47 inhibitor shows promise in high-risk MDS and AML
ENHANCE: Magrolimab does not ameliorate health outcomes in high-risk MDS
Can MRD-guided azacitidine treatment improve outcomes in AML and MDS?
Can WGTS replace standard-of-care diagnostics in AML?
Non-malignant Haematology
ENERGIZE: Mitapivat meets primary efficacy endpoint in thalassaemia
Sovleplenib delivers durable responses and QoL improvements in primary ITP
Avatrombopag successful in children with chronic ITP
RUBY: Promising data for first AsCas12a gene-editing therapy in sickle cell disease
Encouraging data for ELA026 to treat secondary haemophagocytic lymphohistiocytosis
Myelofibrosis
Navitoclax plus ruxolitinib leads to spleen volume reductions in myelofibrosis
Is pelabresib plus ruxolitinib the paradigm-shifting combo therapy for myelofibrosis?
Lymphoma
The landscape of TP53 mutations and their prognostic impact in CLL
Can golcadomide plus R-CHOP become the first-line standard of care in high-risk BCL?
High survival rates following atezolizumab consolidation in DLBCL
First results for zanubrutinib plus venetoclax in del(17p)/TP53-mutated CLL/SLL
EPCORE CLL-1: Promising data for epcoritamab in high-risk Richter’s transformation
Updates from the EBMT Lymphoma Working Group: outcomes after allo- and auto-SCT for T-cell lymphoma subtypes
ECHO: Can we expect a novel standard of care in newly diagnosed MCL?
Clinically meaningful outcomes for mosunetuzumab across follicular lymphoma subgroups
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com


