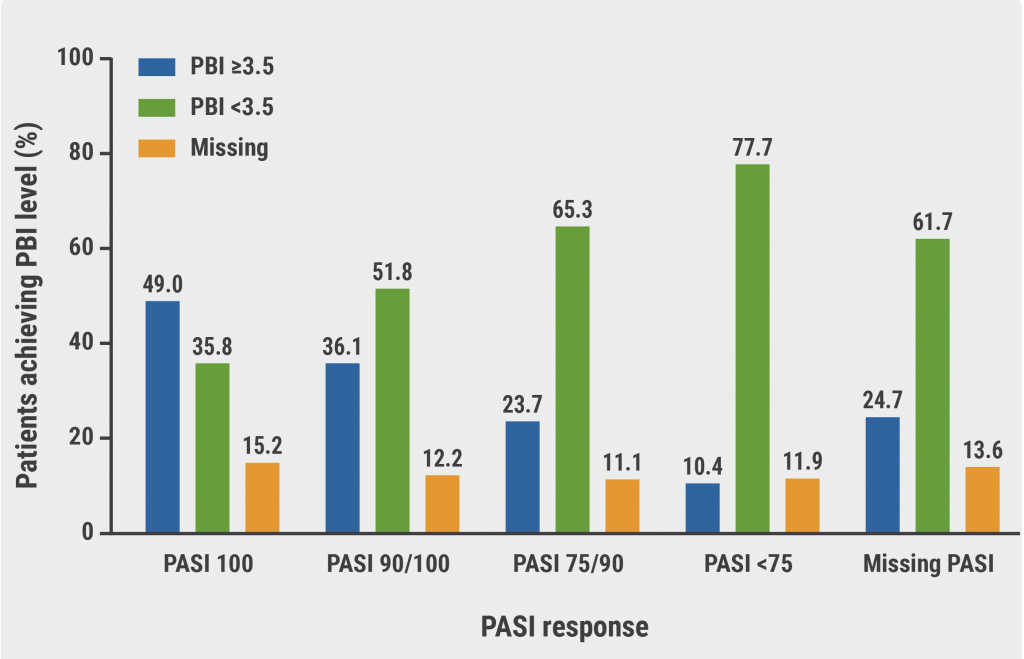
Reasons for prescribing loading doses in biologic treatment are various, including aiming for a faster clinical response and to quickly achieve therapeutic drug levels and disease control at the beginning of treatment [1,2]. Dr Charlotte Greif (Beth Israel Deaconess Medical Center, MA, USA) and her colleagues reviewed 254 studies that compared biologic treatment for psoriasis and other inflammatory diseases, such as rheumatic diseases or inflammatory bowel disease, concerning the advantages of loading doses [1]. Only 5 of those studies included head-to-head assessment of loading doses, with direct comparison data for adalimumab, etanercept, and secukinumab. To evaluate different regimens, maximum dosages over the induction phase (weeks 1–12) and the maintenance period (weeks 13–34) were calculated for each agent and diagnosis. This resulted in a possibility of overall discrimination between high-dose and low-dose treatments.
In general, the systemic review showed higher loading and maintenance dosing in patients treated for dermatologic conditions compared with other inflammatory diseases. This difference was seen for both treatment phases (i.e. induction and maintenance), with the highest quantity of drug administration in patients with hidradenitis suppurativa. The higher dosing in dermatologic diseases could be due to the extension of the involved surface, especially in obese patients.
For psoriasis, the different studies revealed that patients can benefit in the short term when loading doses are administered, leading to faster ameliorations with, for example, greater reductions in Psoriasis Area and Severity Index (PASI). However, this advantage does not seem to be reflected in the long-term efficacy of the treatment. Thus, the value of loading doses may be speed to improvement rather than the overall success rate.
- Greif C, et al. Do loading doses in systemic therapy for psoriasis improve clinical outcomes? P33121, AAD 2022 Annual Meeting, 25–29 March, Boston, MA, USA.
- Geurts-Voerman GE, et al. BMC Rheumatol. 2020 Jul 28;4:37.
Copyright ©2022 Medicom Medical Publishers
Posted on
Previous Article
« JAK inhibitors in AD: Setting the efficacy bar even higher Next Article
Upadacitinib: Fast and more pronounced skin improvement in AD patients »
« JAK inhibitors in AD: Setting the efficacy bar even higher Next Article
Upadacitinib: Fast and more pronounced skin improvement in AD patients »
Table of Contents: AAD 2022
Featured articles
Letter from the Editor
Lebrikizumab treatment leads to encouraging outcomes in multiple traits of AD
New Developments and Unmet Needs in Dermatology
Light at the end of the tunnel for vitiligo therapy
Intestinal microbe-preparation: Modest activity but safe for mild psoriasis
Alopecia areata: 1-year baricitinib treatment increases success
New anticholinergic preparation is effective and tolerable in hyperhidrosis
What’s Hot in Rare Diseases
Add-on apremilast may improve recalcitrant dermatomyositis
Could dupilumab put an end to the therapeutic draught in prurigo nodularis?
Fungal skin infections in children: A diagnosis to keep in mind
Innovative gel speeds up clearance of molluscum contagiosum lesions
JAK inhibition offers promising treatment prospects for uncommon dermatoses
JAK inhibitors may offer a new horizon in the treatment of sarcoidosis
Psoriasis: State of the Art
New insights into psoriasis comorbidity
Long-term psoriasis treatment with bimekizumab results in maintained efficacy
Novel developments in topical psoriasis therapy
Atopic Dermatitis: Novel Agents Enter the Stage
JAK inhibitors in AD: Setting the efficacy bar even higher
Lebrikizumab treatment leads to encouraging outcomes in multiple traits of AD
Novel IL-4/IL-13 blocker shows high efficacy with only modest conjunctivitis signal
Posters
Inpatient dermatologic therapy is linked to lower mortality and readmission rates
AD treatment during the pandemic: dupilumab does not raise COVID-19 infection risk
Upadacitinib: Fast and more pronounced skin improvement in AD patients
Dermatology diseases need the highest doses of biologics
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy