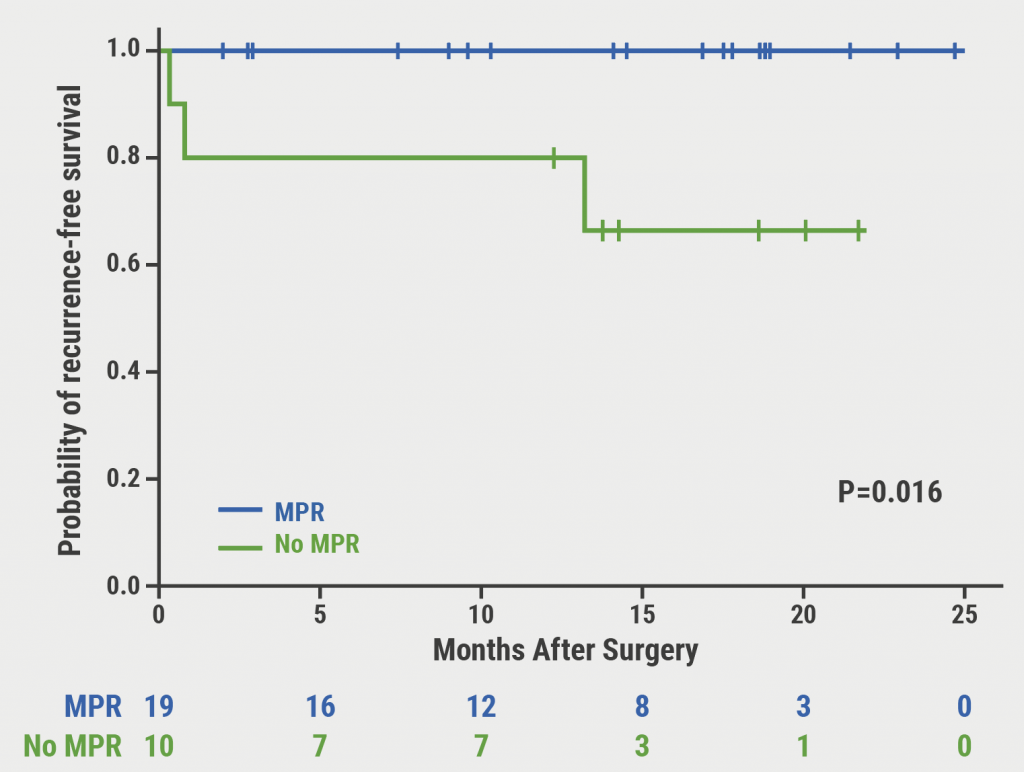
Immune-related adverse events have been reported in up to 80% of patients receiving immune checkpoint inhibitor monotherapy and up to 95% of patients receiving immune checkpoint inhibitor combination therapy [1]. Increasing evidence suggests that the occurrence of immune-related adverse events may be predictive of improved outcomes in cancers such as NSCLC [2]. The phase 3 IMpower130 (NCT02367781), IMpower132 (NCT02657434), and IMpower150 (NCT02366143) trials evaluated efficacy and tolerability of atezolizumab in NSCLC [3-5]. These 3 trials enrolled a total of 2,503 patients with stage IV NSCLC: 1,577 patients were treated with atezolizumab, 926 patients were in the control arms.
A post-hoc pooled analysis, presented by Dr Mark Socinski (AdventHealth Cancer Institute, FL, USA), explored the association between immune-related adverse events and efficacy in these trials [6]. In the atezolizumab arm, 753 patients experienced an immune-related adverse event, and 824 patients were without immune-related adverse events. In the control arm, 289 patients experienced an immune-related adverse event, whereas 637 patients did not. Patients who experienced an immune-related adverse event had a longer overall survival than those without immune-related adverse events in both the atezolizumab and control arm (see Figure). The median overall survival in the atezolizumab arm was 25.7 months versus 13.0 months for patients with versus without immune-related adverse events (HR 0.69; 95% CI 0.60–0.78). In the control arm, median survival was 20.2 months versus 12.8 months, respectively (HR 0.82; 95% CI 0.68–0.99). In the atezolizumab arm, objective response rate was 61.1% in patients who experienced an immune-related adverse event and 37.2% in patients without immune-related adverse events.
Figure: Overall survival stratified by the occurrence of immune-related adverse events [6]

- Jamal S, et al. Rheumatol. 2020;47:1656-175.
- Zhou X, et al. BMC Med. 2020;18:87.
- West H, et al. Lancet Oncol. 2019;20:924-937.
- Nishio M, et al. Thorac Oncol. 2021;16:653-664.
- Socinski MA, et al. N Engl J Med. 2018;378:2288-2301.
- Socinski MA, et al. Pooled analyses of immune-related adverse events (irAEs) and efficacy from the phase 3 trials IMpower130, IMpower132, and IMpower150. Abstract 9002, ASCO 2021 Virtual Meeting, 4–8 June.
Copyright ©2021 Medicom Medical Publishers
Posted on
Previous Article
« Sustained efficacy of nivolumab/ipilimumab plus 2 cycles of chemotherapy in NSCLC Next Article
Novel dual checkpoint blockade improves progression-free survival in melanoma »
« Sustained efficacy of nivolumab/ipilimumab plus 2 cycles of chemotherapy in NSCLC Next Article
Novel dual checkpoint blockade improves progression-free survival in melanoma »
Table of Contents: ASCO 2021
Featured articles
Downloadable 1-Page Editor-Selected Trial PowerPoint Slides
Breast Cancer
Excellent prognosis for breast cancer patients with ultra-low-risk gene signature
Olaparib benefits early breast cancer patients with BRCA1/2 germline mutation
Platinum-based adjuvant chemotherapy in TNBC is not superior or non-inferior to capecitabine
Dalpiciclib benefits patients with HR-positive, HER2-negative advanced breast cancer
Trastuzumab-deruxtecan showed clinical activity in patients with brain metastases
Lung Cancer
Neoadjuvant nivolumab plus chemotherapy improves surgical outcomes in NSCLC
Immune-related adverse events are associated with efficacy of atezolizumab in patients with advanced NSCLC
Sustained efficacy of nivolumab/ipilimumab plus 2 cycles of chemotherapy in NSCLC
Patritumab deruxtecan (HER3-DXd) in EGFR TKI-resistant NSCLC
Melanoma
Long-term results from ground-breaking melanoma trials
Novel dual checkpoint blockade improves progression-free survival in melanoma
Neoadjuvant therapy with nivolumab plus relatlimab is safe and effective in patients with stage III melanoma
Genitourinary Cancers
VISION trial shows improved survival with 177Lu-PSMA-617 in mCRPC
Abiraterone added to ADT + docetaxel nearly doubles survival in de novo mCSPC
Post-nephrectomy pembrolizumab improves disease-free survival
Glutaminase inhibitor telaglenastat does not improve survival mRCC
Promising efficacy and safety of feladilimab in recurrent/metastatic urothelial carcinoma
Gastrointestinal Cancers
Pembrolizumab benefits survival in MSI-H/dMMR metastastic colorectal cancer
Panitumumab added to 5-FU/LV effective as maintenance therapy in patients with mCRC
Trastuzumab-deruxtecan showed promising activity in patients with HER2-expressing mCRC
Benefit of both I-O/chemo combo and I-O/I-O combo over chemotherapy alone in oesophageal squamous cell cancer
Benefit of I-O/chemo combo over chemotherapy alone in advanced GC/GEJC/EAC
Perioperative chemotherapy and neoadjuvant multimodality therapy appear equally effective
Haematological Cancers
Olutasidenib demonstrates efficacy in patients with relapsed/refractory IDH1 mutant AML
Acalabrutinib as effective but better tolerated than ibrutinib in CLL
Gynaecological Cancers
Adjuvant chemotherapy does not improve outcome in patients with locally advanced cervical cancer
Novel drug combination for recurrent ovarian cancer
Dual HER2-blockade shows anti-tumour activity in patients with uterine cancer
Paediatric Cancer
Molecular tumour profiling impacts the diagnosis and treatment of solid tumours
Circulating tumour DNA to evaluate response in children with neuroblastoma
Basic Science
PARP7 inhibitor shows promising results in first-in-human trial
IACS-6274 is well tolerated and biologically active in selected advanced tumours
CYT-0851 shows promising anti-tumour activity across different tumour types
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com