https://doi.org/10.55788/b17d8dab
Central neuropathic pain is a complex problem and experienced differently from patient to patient. It is common in patients with e.g. MS or spinal cord injury, it severely affects quality-of-life, and is often difficult to treat. CBM has been suggested as a treatment option for a wide range of diseases and symptoms, including central neuropathic pain. The Danish Sclerosis Association has requested CBM, due to patients having high expectations of effects and of a favourable safety profile compared with the many non-medical-grade products chronic patients already use. Over 125 different cannabinoids are known to exist. Of specific medical interest are Δ9-tetrahydrocannabinol (THC), which is psychotropic, and cannabidiol (CBD), which is not. Animal studies have shown promising results of CBM, but it has been difficult to confirm a clear effect of CBM on pain in humans. A meta-analysis evaluating isolated CBD for pain is lacking.
This Danish multicentre study, presented by Dr Julie Hansen (Aarhus University, Denmark), aimed to evaluate the effect of CBM on patient-reported central neuropathic pain and/or spasticity in a population of patients with MS or spinal cord injury [1,2]. The study had a randomised, double-blind, placebo-controlled, parallel design. Study medication consisted of oral capsules of THC (natural), CBD (synthetic), or a combination (THC&CBD), at a maximum daily dose of 22.5 mg, 45 mg, and 22.5+45 mg, respectively. During 7 weeks of treatment and 4 weeks of follow-up, pain intensity was registered daily using a Numeric Rating Scale (NRS, 0–10).
489 patients were assessed for eligibility with central neuropathic pain due to MS or spinal cord injury, of whom 114 with central neuropathic pain >3 were included; mean age was 52 years and 77% were women. The mean difference in pain intensity from baseline to the last week of treatment was 1.8 for placebo (n=35; 95% CI 1.17–2.43), 1.4 for THC (n=24; 95% CI 0.53–2.24), 1.4 for CBD (n=27; 95% CI 0.72–1.98), and 1.6 for THC&CBD (n=28; 95% CI 0.94–2.34). There was no significant difference in pain reduction between the groups. The effect sizes of THC, CBD, and THC&CBD were 0.42, 0.45, and 0.16, respectively (P=0.74, one-way ANOVA). The effect size of the isolated cannabinoids was 0.83 for THC, and 0.71 for CBD (P=0.31). Pain intensity was lowered in all 4 groups, and regressed to baseline values during the 4 weeks of follow-up.
In conclusion, CBM did not prove more effective than a placebo in the treatment of chronic neuropathic pain. As all groups reported a lower pain intensity, this study suffered from a high placebo effect.
- Hansen JS, et al. The effect of cannabis-based medicine on central neuropathic pain – results from a randomized, placebo-controlled trial. EAN 2023 Annual Meeting, 1–4 July, Budapest, Hungary.
- Hansen JS, et al. Brain Sci. 2021;11(9):1212.
Copyright ©2023 Medicom Medical Publishers
Posted on
Previous Article
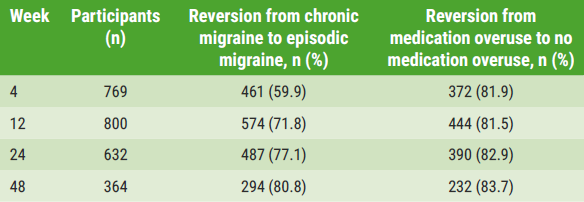
« 80% of patients reverse from chronic to episodic migraine on anti-CGRP antibodies Next Article
GLP-1 agonists induce weight loss and alleviate headache in idiopathic intracranial hypertension »
« 80% of patients reverse from chronic to episodic migraine on anti-CGRP antibodies Next Article
GLP-1 agonists induce weight loss and alleviate headache in idiopathic intracranial hypertension »
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com