Homologous recombination is an essential, high-fidelity mechanism to repair DNA double-strand breaks. Overexpression of the cytidine deaminase family of DNA damaging enzymes in subsets of cancers leads to increased DNA damage and subsequent dependence on homologous recombination-mediated DNA repair. Inhibition of homologous recombination in cancer cells leads to accumulation of unrepaired DNA double-strand breaks and tumour cell death. RAD51 is a key enzyme involved in homologous recombination. CYT-0851 is an oral, first-in-class, small-molecule inhibitor of RAD51-mediated DNA repair. Previously, CYT-0851 showed anticancer activity in preclinical models of solid and haematological cancer [1,2].
Dr Ryan Lynch (Fred Hutchinson Cancer Research Center, WA, USA) presented the first results of a phase 1-2 trial (NCT03997968) of CYT-0851 in patients with advanced solid and haematologic cancers [3]. Primary objectives included safety, maximum tolerated dose, recommended phase 2 dose, and anti-tumour activity. Secondary and exploratory objectives included pharmacokinetics, pharmacodynamics, and predictive biomarkers of response.
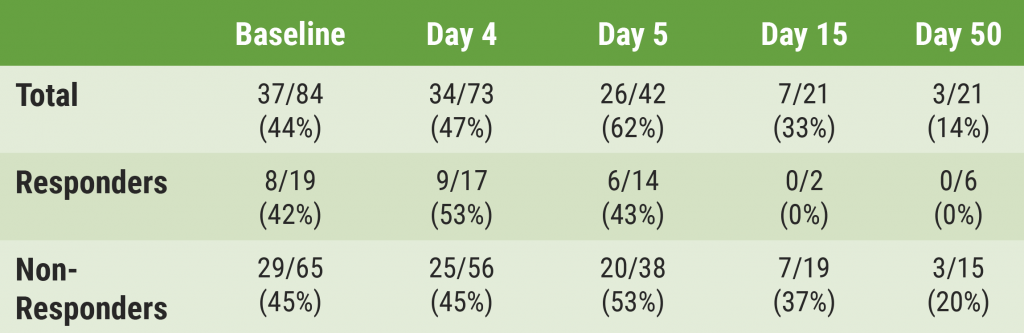
At data cut-off, 39 patients were enrolled of whom 35 were treated with CYT-0851. Diagnoses were sarcoma (n=12), non-Hodgkin lymphoma (n=8), breast cancer (n=5), pancreatic cancer (n=4), ovarian cancer (n=3), head and neck cancer (n=1), small cell lung cancer (n=1), and mucoepidermoid carcinoma (n=1). Dosing ranged from 15–45 mg twice daily to 90–3,000 mg once daily.
No patients experienced dose-limiting toxicity. A total of 13 patients (37%) experienced a CYT-0851-related adverse event with only 1 patient experiencing a grade ≥3 adverse event (fatigue). No clinically significant myelosuppression or treatment-related discontinuation was observed. Preliminary pharmacokinetic analyses showed dose-proportional systemic exposure with a half-life of ~3 days, supporting the transition from twice-daily to once-daily dosing.
A total of 21 patients were response-evaluable; 3 partial responses were observed (2 patients with non-Hodgkin lymphoma, 1 patient with soft-tissue sarcoma), and 10 patients had stable disease. Dose escalation continues to establish the recommended phase 2 dose, with planned expansion in 7 disease-specific cohorts in haematological and solid cancers.
- Day M, et al. Cancer Res. 2019;79(suppl):C14.
- Day M, et al. Blood 2019;134(suppl 1):2080.
- Lynch RC, et al. First-in-human phase I/II study of CYT-0851, a first-in-class inhibitor of RAD51-mediated homologous recombination in patients with advanced solid and hematologic cancers. Abstract 3006, ASCO 2021 Virtual Meeting, 4-8 June.
Copyright ©2021 Medicom Medical Publishers
Posted on
Previous Article
« Acalabrutinib as effective but better tolerated than ibrutinib in CLL Next Article
Icotinib tops chemo as adjuvant therapy for EGFR-mutant NSCLC »
« Acalabrutinib as effective but better tolerated than ibrutinib in CLL Next Article
Icotinib tops chemo as adjuvant therapy for EGFR-mutant NSCLC »
Table of Contents: ASCO 2021
Featured articles
Downloadable 1-Page Editor-Selected Trial PowerPoint Slides
Breast Cancer
Excellent prognosis for breast cancer patients with ultra-low-risk gene signature
Olaparib benefits early breast cancer patients with BRCA1/2 germline mutation
Platinum-based adjuvant chemotherapy in TNBC is not superior or non-inferior to capecitabine
Dalpiciclib benefits patients with HR-positive, HER2-negative advanced breast cancer
Trastuzumab-deruxtecan showed clinical activity in patients with brain metastases
Lung Cancer
Neoadjuvant nivolumab plus chemotherapy improves surgical outcomes in NSCLC
Immune-related adverse events are associated with efficacy of atezolizumab in patients with advanced NSCLC
Sustained efficacy of nivolumab/ipilimumab plus 2 cycles of chemotherapy in NSCLC
Patritumab deruxtecan (HER3-DXd) in EGFR TKI-resistant NSCLC
Melanoma
Long-term results from ground-breaking melanoma trials
Novel dual checkpoint blockade improves progression-free survival in melanoma
Neoadjuvant therapy with nivolumab plus relatlimab is safe and effective in patients with stage III melanoma
Genitourinary Cancers
VISION trial shows improved survival with 177Lu-PSMA-617 in mCRPC
Abiraterone added to ADT + docetaxel nearly doubles survival in de novo mCSPC
Post-nephrectomy pembrolizumab improves disease-free survival
Glutaminase inhibitor telaglenastat does not improve survival mRCC
Promising efficacy and safety of feladilimab in recurrent/metastatic urothelial carcinoma
Gastrointestinal Cancers
Pembrolizumab benefits survival in MSI-H/dMMR metastastic colorectal cancer
Panitumumab added to 5-FU/LV effective as maintenance therapy in patients with mCRC
Trastuzumab-deruxtecan showed promising activity in patients with HER2-expressing mCRC
Benefit of both I-O/chemo combo and I-O/I-O combo over chemotherapy alone in oesophageal squamous cell cancer
Benefit of I-O/chemo combo over chemotherapy alone in advanced GC/GEJC/EAC
Perioperative chemotherapy and neoadjuvant multimodality therapy appear equally effective
Haematological Cancers
Olutasidenib demonstrates efficacy in patients with relapsed/refractory IDH1 mutant AML
Acalabrutinib as effective but better tolerated than ibrutinib in CLL
Gynaecological Cancers
Adjuvant chemotherapy does not improve outcome in patients with locally advanced cervical cancer
Novel drug combination for recurrent ovarian cancer
Dual HER2-blockade shows anti-tumour activity in patients with uterine cancer
Paediatric Cancer
Molecular tumour profiling impacts the diagnosis and treatment of solid tumours
Circulating tumour DNA to evaluate response in children with neuroblastoma
Basic Science
PARP7 inhibitor shows promising results in first-in-human trial
IACS-6274 is well tolerated and biologically active in selected advanced tumours
CYT-0851 shows promising anti-tumour activity across different tumour types
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com