Chronic rhinosinusitis with nasal polyps (CRSwNP) involves chronic inflammation of the rhinosinal cavities, mostly of type 2 signature, combined with growth of polyps in the nasal airway [2,3]. CRSwNP, entailing symptoms like congestion, facial pain, and hyposmia, also negatively impacts quality of life [2,4,5]. The current standard of care relies on intranasal corticosteroids (CS) with short courses of systemic CS if needed and nasal surgery in case of CS failure [3]. “Unfortunately, the benefit of this is often short-lived, recurrences are common, and many patients suffer from ongoing inadequate control of their disease and symptoms,” said Dr Claire Hopkins (Guy’s and St Thomas Hospital and King’s College London, UK).
Previous investigations with 750 mg mepolizumab IV detected an improvement of CRSwNP with this IL-5 antibody [6,7]. The current phase 3, randomised, placebo-controlled SYNAPSE trial evaluated subcutaneous mepolizumab at a dose of 100 mg every 4 weeks over 52 weeks as add-on to standard of care (i.e. mometasone furoate plus saline nasal douching) for adult patients with CRSwNP. The 407 participants had at least 1 previous surgery and were deemed to need further surgery. The mean age was 49 years, mean duration of nasal polyposis was 11.45 years, and 38% in the placebo versus 33% in the mepolizumab group were female. Rescue treatment with oral prednisolone antibiotics or surgery was permitted. The co-primary endpoints were defined as change from baseline in endoscopic nasal polyp score at week 52 in addition to change from baseline in nasal obstruction in visual analogue scale (VAS) score during weeks 49-52. The key secondary endpoint consisted of time-to-first actual nasal surgery up to week 52.
“More than 50% of patients achieved significant reduction in nasal polyp score with some patient achieving a greater than 5-point score reduction,” Dr Hopkins revealed. The median difference in total endoscopic nasal polyp score was -0.73 (95% CI -1.11 to -0.34; P<0.001) (see Figure). In the nasal obstruction VAS score, 44% of participants achieved a reduction of >5 points in the mepolizumab group compared with 23% in the placebo group (see Figure). With reference to surgery, only 9% of the patients with add-on mepolizumab underwent surgery versus 23% receiving placebo.
Figure: Co-primary endpoints of the SYNAPSE trial [1]
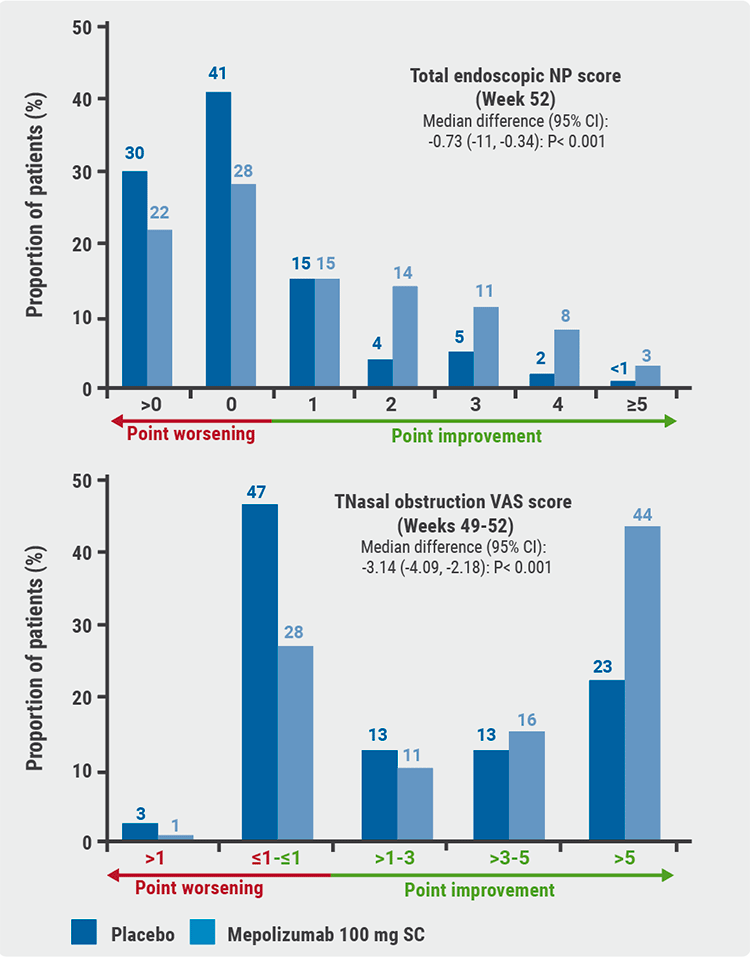
NP, nasal polyps; VAS, visual analogue scale.
In terms of safety, on-treatment adverse events were reported by 82% of the mepolizumab participants and 84% by the participants in the placebo group. Serious adverse events were noted in <1% on placebo and 6% on mepolizumab, but these were rated as unelated to study treatment.
“In conclusion, we demonstrated in the SYNAPSE study that add-on mepolizumab is an effective and safe treatment for adults with chronic rhinosinusitis with nasal polyps,” Dr Hopkins concluded her talk.
For more articles on Type 2 Inflammation, see our independent webportal inflammation-type2.org.
- Hopkins C. Add-on mepolizumab for chronic rhinosinusitis with nasal polyps: SYNAPSE study. LBA 4616, ERS International Virtual Congress 2020, 7-9 Sept.
- Stevens WW, et al. J Allergy Clin Immunol Pract. 2016;4:565-572.
- Fokkens WJ, et al. Allergy. 2019;74:2312-2319.
- Schleimer RP, et al. Annu Rev Pathol. 2017;12:331-357.
- Kim J, et al. Ther Clin Risk Manag. 2020;16:31-37.
- Bachert C, et al. J Allergy Clin Immunol. 2017;140:1024-1031.
- Gevaert P, et al. J Allergy Clin Immunol. 2011;128:989-995.
Posted on
Previous Article
« COVID-19 survivors benefit from structured follow-up Next Article
Secondary pulmonary fibrosis: a possible long-term effect of severe COVID-19 »
« COVID-19 survivors benefit from structured follow-up Next Article
Secondary pulmonary fibrosis: a possible long-term effect of severe COVID-19 »
Table of Contents: ERS 2020
Featured articles
COVID-19 and the Lung
COVID-19 infections: Bronchoscopy provides additional diagnostic certainty
COVID-19 vaccines: An ongoing race
COVID-19: What is the risk of reinfection?
COVID-19 App: The Dutch experience
Secondary pulmonary fibrosis: a possible long-term effect of severe COVID-19
COVID-19 survivors benefit from structured follow-up
Early pulmonary rehabilitation post-COVID-19 aids recovery
Asthma – What's New
Mild asthma: A fundamental change in management
Dupilumab shows long-term efficacy in asthma patients
Severe asthma: Oral corticosteroids maintenance therapy associated with toxicity
First-in-class tyrosine kinase inhibitor shows promise in severe asthma
Predicting individual effectiveness of biologics in severe asthma
IL-5 antagonist showed efficacy in chronic rhinosinusitis with nasal polyps
Treatment according to genotype: The future of asthma therapy?
COPD – The Beat Goes On
The role of chronic symptoms as early biomarkers of COPD development
Urgent call for studies in COPD patients aged 40-60 years
Nasal high-flow therapy: a novel treatment option for hypercapnic COPD patients
Exacerbation history is a reliable predictor of future exacerbations
Singing training effective as physical rehabilitation in COPD
Current prediction tools underestimate exacerbation risk of severe COPD patients
Exercise and Sleep: From Impaired Function to New Therapeutic Strategies
CPAP withdrawal has negative consequences for sleep apnoea patients
Physical activity improves AHI in sleep apnoea patients
The Tobacco Epidemic: From Vaping to Cannabis
Poly-use of nicotine products and cannabis: a deadly combination
E-cigarettes: A source of chronic lung inflammation
Social smoking: Do not underestimate the risks
Chronic Cough – State of the Art
LEAD study shows multiple phenotypes in many chronic cough patients
First-in-class P2X3 receptor antagonist shows promise for chronic cough treatment
Lung Cancer Detection
Lung cancer screening: Most patients not eligible 1-2 years prior to diagnosis
Distinct changes in lung microbiome precede clinical diagnosis of lung cancer
Best of Posters
Smartphone-based cough detection helpful in predicting asthma deterioration
Reduced lung function associated with cognitive decline in the elderly
Longer hospital stay and fewer transplants for frail ILD patients
Related Articles

September 22, 2022
ERS 2022 Highlights Podcast
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy
HEAD OFFICE
Laarderhoogtweg 25
1101 EB Amsterdam
The Netherlands
T: +31 85 4012 560
E: publishers@medicom-publishers.com

