Trastuzumab deruxtecan is a HER2-targeting, antibody-drug conjugate for the treatment of previously treated patients with advanced HER2-positive metastatic breast cancer, as was demonstrated by the DESTINY-Breast01 study (NCT03248492) [1]. Before, the EMILIA trial (NCT00829166) showed trastuzumab emtansine to be beneficial in this population [2]. In the phase 3 DESTINY-Breast03 (NCT03529110) trial the efficacy and safety of trastuzumab deruxtecan and trastuzumab emtansine are compared head-to-head in patients previously treated with trastuzumab and taxane. The primary analysis demonstrated trastuzumab deruxtecan to have a clinically meaningful and statistically significant improved PFS versus trastuzumab emtansine. Trastuzumab deruxtecan demonstrated superior PFS versus trastuzumab emtansine (HR 0.28; P<0.0001); median PFS was not reached for trastuzumab deruxtecan versus 6.8 months for trastuzumab emtansine; 12-month PFS rate was 79.7% versus 34.2%, respectively [3]. Now, Dr Sara Hurvitz (University of California, CA, USA) presented results from subgroup analyses of DESTINY-Breast03, including patients with brain metastases [4].
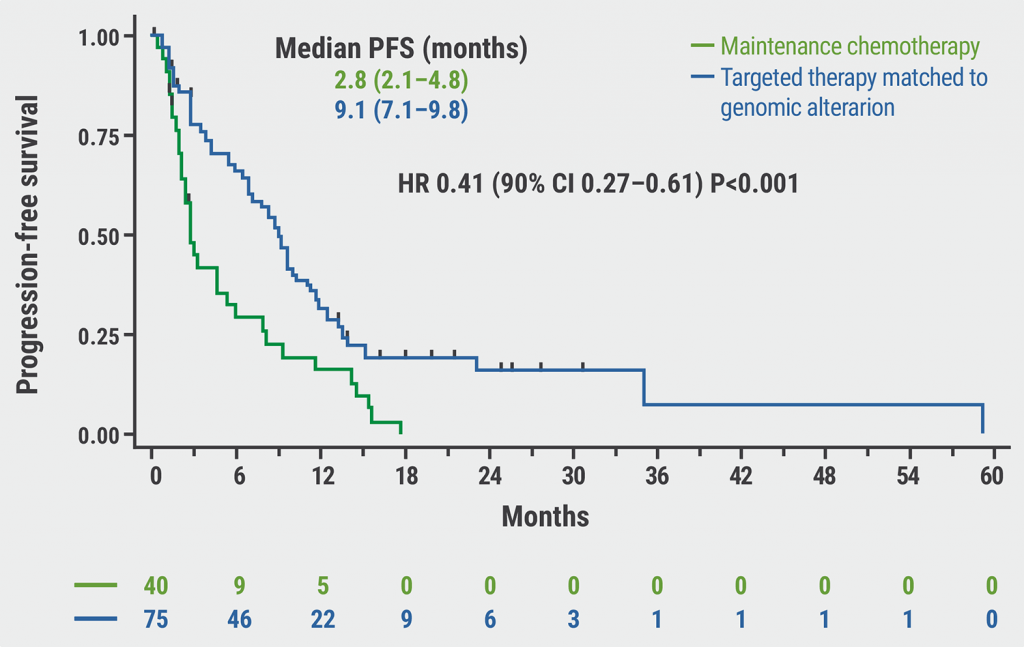
Median PFS favoured trastuzumab deruxtecan over trastuzumab emtansine independent of hormone receptor status, prior trastuzumab treatment, and number of prior lines of therapy. For patients with stable brain metastases at baseline (n=82), median PFS was 15.0 months for trastuzumab deruxtecan versus 3.0 months for trastuzumab emtansine (HR 0.25; see Figure). Overall, confirmed overall response rate (ORR) for trastuzumab deruxtecan was 79.7% (16.1% complete response, 63.6% partial response) versus 34.2% (8.2% complete response, 25.5% partial response) for trastuzumab emtansine. In patients with stable brain metastases at baseline, ORR was 67.4% (4.7% complete response, 62.8% partial response) for trastuzumab deruxtecan versus 20.5% (0% complete response, 20.5% partial response) for trastuzumab emtansine. In addition, intracranial response rate in these patients was 63.9% (27.8% complete response, 36.1% partial response) for trastuzumab deruxtecan versus 33.4% (2.8% complete response, 30.6% partial response) for trastuzumab emtansine.
Figure: Progression-free survival of patients with and without brain metastases treated with trastuzumab deruxtecan or trastuzumab emtansine [4].

mPFS, median progression-free survival; PFS, progression-free survival.
Overall, the safety profile of trastuzumab deruxtecan was manageable and comparable with its known safety profile. Adjudicated drug-related interstitial lung disease/pneumonitis was reported in 27 (10.5%) patients treated with trastuzumab deruxtecan and 5 (1.9%) patients treated with trastuzumab emtansine, with no grade 4 or 5 events.
“Consistent PFS and ORR benefits with trastuzumab deruxtecan versus trastuzumab emtansine were observed across subgroups in patients with HER2-positive metastatic breast cancer previously treated with trastuzumab and taxanes, including patients with brain metastases,” concluded Dr Hurvitz.
- Modi S, al. N Engl J Med 2020;382:610–621.
- Verma S, Miles D, et al. N Engl J Med 2012; 367:1783-1791.
- Cortés J, et al. Ann Oncol. 2021;32(suppl5):1283–1346.
- Hurvitz S, et al. Trastuzumab deruxtecan (T-DXd; DS-8201a) vs. trastuzumab emtansine (T-DM1) in patients (pts) with HER2+ metastatic breast cancer (mBC): subgroup analyses from the randomized phase 3 study DESTINY-Breast03. GS3-01, SABCS 2021 Virtual Meeting, 7–10 December.
Copyright ©2022 Medicom Medical Publishers
Posted on
Previous Article
« Nivolumab plus ipilimumab serve promising dual checkpoint inhibition Next Article
Datopotamab deruxtecan shows promising anti-tumour activity »
« Nivolumab plus ipilimumab serve promising dual checkpoint inhibition Next Article
Datopotamab deruxtecan shows promising anti-tumour activity »
Table of Contents: SABCS 2021
Featured articles
Early-Stage Breast Cancer
Aromatase inhibitors outperform tamoxifen in premenopausal women
Concurrent taxane plus anthracycline most beneficial in reducing risk of breast cancer
Reduced risk of recurrence with ovarian suppression plus tamoxifen/exemestane
Metformin does not improve outcomes in patients with early-stage breast cancer
Omitting sentinel lymph node biopsy improves arm symptoms
HR-positive/HER2-negative Breast Cancer
Addition of palbociclib to standard endocrine therapy does not improve outcome in adjuvant treatment
The SERD elacestrant improves outcomes for patients unresponsive to endocrine therapy
Consistent overall survival benefit of ribociclib in advanced breast cancer
Premenopausal women benefit from adjuvant chemotherapy next to endocrine therapy
Promising anti-tumour activity of the CDK7-inhibitor samuraciclib plus fulvestrant
ctDNA is prognostic and predictive for response to ribociclib plus letrozole
Early switch to fulvestrant plus palbociclib beneficial for patients with ESR1 mutation
Triple-Negative Breast Cancer
Single-cell spatial analysis can predict response to neoadjuvant immunotherapy
Neoadjuvant pembrolizumab plus chemotherapy benefits event-free survival in TNBC
Early use of ctDNA testing can identify likelihood of relapse in TNBC
Pembrolizumab plus chemotherapy benefits patients with combined positive score ≥10
Neratinib plus trastuzumab plus fulvestrant shows encouraging clinical activity
Phase 1–3 Trials
Datopotamab deruxtecan shows promising anti-tumour activity
Trastuzumab deruxtecan outperforms trastuzumab emtansine
Nivolumab plus ipilimumab serve promising dual checkpoint inhibition
Entinostat plus exemestane improves progression-free survival in Chinese patients
Efficacy of pyrotinib plus capecitabine confirmed in previously treated patients
Basic and Translational Research
Using genomics to match treatments improves outcomes
Loss of ASXL1 tumour suppressor promotes resistance to CDK4/6 inhibitors
Inducers of ferroptosis are potential drugs to target p53-mutated TNBC cells
MAPK-pathway alterations are associated with resistance to anti-HER2 therapy
Genomic signatures of DCIS define biology and correlate with clinical outcomes
BRCA2 linked to inferior outcomes with CDK4/6 inhibitors plus endocrine therapy
Miscellaneous
Olaparib is well tolerated as an additional treatment
Race effects the likelihood to develop lymphoedema following breast cancer treatment
Sentinel lymph node staging is non-inferior to complete axillary lymph node dissection
One in 7 breast cancers detected during screening are overdiagnosed
Related Articles
January 31, 2022
Using genomics to match treatments improves outcomes
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy

