Axi-cel and tisa-cel are autologous anti-CD19 CAR T-cell therapies approved in the US and EU for adults with relapsed/refractory (R/R) DLBCL after ≥2 lines of systemic therapy. Dr Emmanuel Bachy (Hospices Civils de Lyon, France) and others conducted a propensity score-matched comparison of axi-cel and tisa-cel in a large cohort of R/R DLBCL patients treated outside of clinical trials. After a 1:1 ratio propensity score-matching, therapy outcomes were compared between 144 patients treated with axi-cel and 144 patients treated with tisa-cel. The primary endpoint was overall survival (OS).
After a median follow-up of 6.6 months, OS was not significantly different between axi-cel and tisa-cel at 6 months (78% vs 70% respectively; P=0.44; see Figure). Best overall and complete response rates were significantly higher with axi-cel compared with tisa-cel (73% vs 60%; P=0.02; and 56% vs 36%; P<0.001, respectively). At 6 months, PFS was significantly longer with axi-cel than with tisa-cel (53% vs 32%; P=0.011).
Figure: Overall survival with tisa-cel versus axi-cel [1]
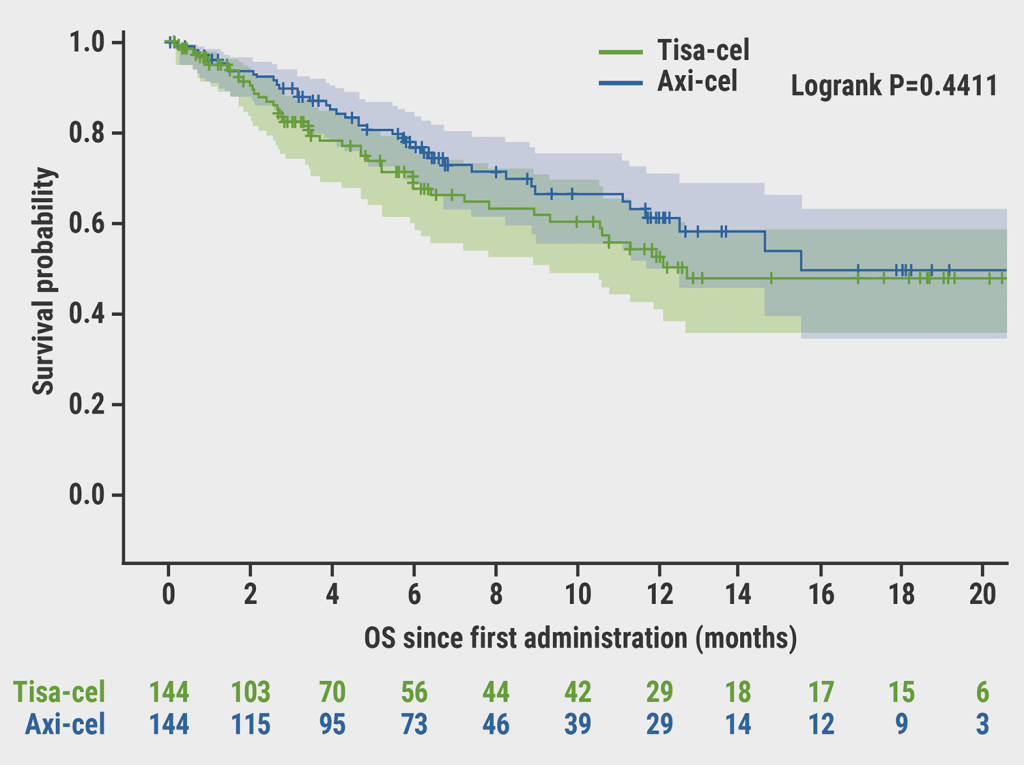
Axi-cel, axicabtagene ciloleucel; OS, overall survival; tisa-cel, tisagenlecleucel.
With respect to the toxicity profile, there was no significant difference in the incidence of cytokine release syndrome (CRS), but axi-cel was associated with significantly more frequent neurotoxicity (ICANS) compared with tisa-cel, namely:
- 6% vs 18.1% for grade 1–2 ICANS; and
- 4% vs 2.1% for grade ≥3 ICANS (P<0.001).
After stringent propensity score-matching on a large patient population treated with CAR T-cell therapy, axi-cel resulted in higher response rates and significantly prolonged PFS compared with tisa-cel. However, greater efficacy came at the cost of higher neurotoxicity with axi-cel.
- Bachy E, et al. A Propensity Score-Matched Comparison of Axi-Cel and Tisa-Cel for Relapsed/Refractory Diffuse Large B-Cell Lymphoma in Real-Life: A Lysa Study from the Descar-T Registry. Abstract 92, ASH 2021 Annual Meeting, 11–14 December.
Copyright ©2022 Medicom Medical Publishers
Posted on
Previous Article
« POLARIX: Novel regimen superior to R-CHOP in DLBCL Next Article
Novel anti-CD19 plus lenalidomide prolonged survival in R/R DLBCL »
« POLARIX: Novel regimen superior to R-CHOP in DLBCL Next Article
Novel anti-CD19 plus lenalidomide prolonged survival in R/R DLBCL »
Table of Contents: ASH 2021
Featured articles
Acute Lymphoblastic Leukaemia
New Interfant protocol includes blinatumomab for KMT2A-r ALL
Persistent disparities in ALL health outcomes
EWALL-INO: Inotuzumab ozogamicin promising as first-line therapy for BCP-ALL
UKALL 2003: Therapy de-escalation safe in low-risk MRD patients with ALL
Acute Myeloid Leukaemia
AMLSG 16-10: Long-term benefits of midostaurin for FLT3-ITD-mutated AML
Comparable effectiveness of CPX-351 and venetoclax plus HMA in older AML patients
Promising frontline triplet regimen for TP53-mutated AML
Encouraging results of novel triplet combination for AML
Heavily pre-treated FLT3-mutated AML population may benefit from novel triplet regimen
Benefits of eprenetapopt plus azacitidine for TP53-mutant MDS and oligoblastic AML
Improved risk stratification in MDS via gene-based scoring system
Chronic Leukaemia
CAPTIVATE: Ibrutinib plus venetoclax shows ongoing efficacy in CLL
SEQUOIA: Zanubrutinib meets primary endpoint for treatment-naïve CLL/SLL
Investigational therapies superior to standard-of-care in double-exposed CLL
Multiple Myeloma
GRIFFIN: Sustained responses of daratumumab plus RVd in MM
MajesTEC-1: Teclistamab efficacious in heavily pre-treated MM
iStopMM: Smouldering MM highly prevalent in general population
Mechanisms of D-KRd treatment failure in MM identified
TRIMM-2: Favourable results of talquetamab plus daratumumab for MM
Lymphoma
Second-line tisa-cel similar to standard-of-care for R/R aggressive non-Hodgkin lymphoma
Axi-cel improved event-free survival in R/R DLBCL
Axi-cel more effective but tisa-cel less toxic in DLBCL
POLARIX: Novel regimen superior to R-CHOP in DLBCL
Novel non-invasive biomarker ctDNA shows value in CNS lymphoma
Myeloproliferative Neoplasms
Mechanisms behind TP53 mutations revealed in myeloproliferative neoplasms
JAK2V617F variant allele frequency prognostic of venous events in polycythaemia vera
Immune Thrombocytopenia
Promising results of tacrolimus plus dexamethasone for ITP
Sustained remission after TPO-RA discontinuation in chronic ITP
Haemophilia
Fitusiran meets primary endpoint in ATLAS-A/B trial
ATLAS-INH: Impressive results of fitusiran for haemophilia with inhibitors
rFVIIIFc establishes rapid tolerisation in haemophilia A with inhibitors
Clonal Haematopoiesis
Reduced risk of Alzheimer’s disease in CHIP carriers
Lifelong patterns of clonal haematopoiesis revealed
Related Articles
February 4, 2022
Reduced risk of Alzheimer’s disease in CHIP carriers
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy

