https://doi.org/10.55788/2681d3a9
“Patients with HFrEF and low SBP are at increased risk of clinical events and do not tolerate guideline-directed medical therapy well,” explained Prof. Marco Metra (University of Brescia, Italy) [1]. Omecamtiv mecarbil is a selective cardiac myosin activator that improves cardiac function, without reducing blood pressure and might benefit these patients with HFrEF and low SBP. Prof. Metra and colleagues analysed the efficacy and safety of omecamtiv mecarbil in patients with HFrEF and low SBP (≤100 mmHg) in the phase 3 GALACTIC-HF trial (NCT02929329; total n=8,232; low SBP n=1,473). The primary analysis showed a significant benefit of omecamtiv mecarbil over placebo in time-to-first HF event or cardiovascular death in the overall population (HR 0.92; P=0.025) [2].
The current subanalysis showed that participants with a low SBP had a higher risk of HF events than participants with an SBP >100 mmHg (HR 1.05 per 5 mmHg; P<0.001). However, the treatment effect of omecamtiv mecarbil on cardiovascular death or time-to-first HF event was larger in those with a low SBP (HR 0.81) than in those with a higher SBP (HR 0.95; Pinteraction=0.051; see Figure). The safety profiles of omecamtiv mecarbil and placebo were similar in both SBP groups. Thus, the authors concluded that the challenging population of patients with HFrEF and low SBP may benefit from treatment with omecamtiv mecarbil.
Figure: Kaplan Meier curves for the primary endpoint by SBP subgroups [1]
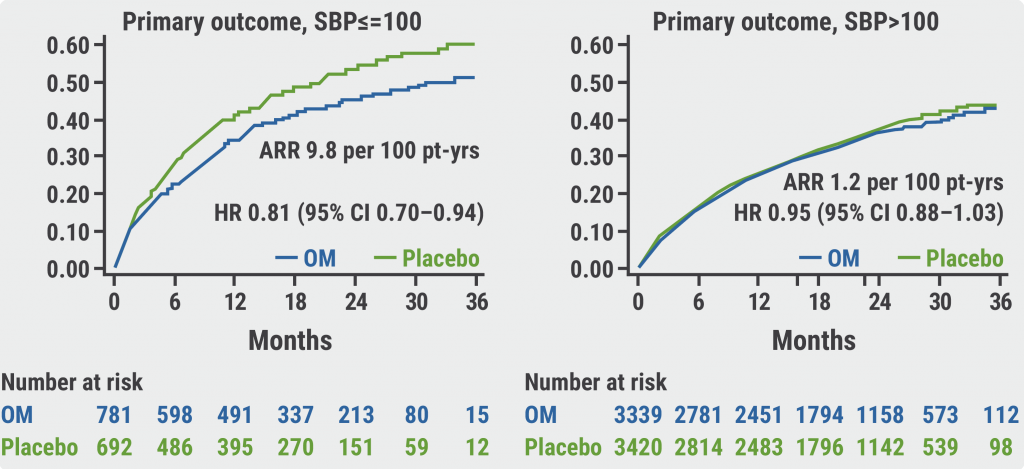
SBP, systolic blood pressure; ARR, absolute risk reduction; OM, omecamtiv mecarbil; HR, hazard ratio.
- Metra M, et al. GALACTIC-HF – omecamtiv mecarbil in HFrEF and low blood pressure. LBT Pharmacological treatment I, Heart Failure 2022, 21–24 May, Madrid, Spain.
- Teerlink JR, et al. JACC Heart Fail. 2020;8:329–340.
Copyright ©2022 Medicom Medical Publishers
Posted on
Previous Article
« HELIOS-A: Vutrisiran meets exploratory endpoints Next Article
Letter from the Editor »
« HELIOS-A: Vutrisiran meets exploratory endpoints Next Article
Letter from the Editor »
Table of Contents: HFA 2022
Featured articles
Phase 3 and 4 Trials
GALACTIC-HF: Omecamtiv mecarbil as option for HFrEF patients with low SBP
HELIOS-A: Vutrisiran meets exploratory endpoints
Patiromer helps HFrEF patients to optimise RAAS inhibitors without hyperkalaemia
FIDELITY: Cardiorenal benefits of finerenone, regardless of LVH status
DAPA-VO2: Rapid effect of dapagliflozin on peak VO2 in stable HFrEF
Phase 1/2 Trials
Significant improvement in BP from istaroxime, a novel non-adrenergic agent
SERENADE: Macitentan fails in HFpEF plus PAH
Combination of filgrastim and dutogliptin appears safe in STEMI
Therapeutic Devices
Cardiac contractility modulation therapy promising for patients with HFpEF
REBALANCE-HF: Encouraging observations for splanchnic nerve ablation in HFpEF
Updates on SGLT2 Inhibitors
DAPA-HF: Dapagliflozin is safe and efficacious in frail patients
EMPEROR-Preserved: Empagliflozin stable across age groups
EMPULSE: Empagliflozin delivers rapid and clinically meaningful decongestion
Dapagliflozin performs consistently across LVEF in HF
Miscellaneous Topics
Cardiac wasting relevant for clinical outcomes in cancer
Urocortin-2 a potential treatment target for HFpEF
Should ATTR-CM be added to the differential diagnosis of patients with HF?
Delayed initiation of novel GDMTs associated with adverse outcomes in HF patients
Related Articles
June 15, 2022
Surprise outcome for SODIUM-HF
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy

