https://doi.org/10.55788/7f2c35ab
The previously published, multicentre, randomised-controlled VANISH trial (NCT00905853) included 259 patients with an implantable cardioverter-defibrillator (ICD) who experienced a prior MI and VT event on AAD treatment in the last 6 months. These patients were randomised to catheter ablation (n=132) or escalated AAD therapy (n=127). After a median follow-up of 23.4 months, the primary composite endpoint of all-cause mortality, VT storm, and appropriate ICD shock favoured ablation over escalated AAD therapy (HR 0.72; P=0.04) [1]. In the current analysis, Prof. Michelle Samuel (University of Montreal, Canada) and colleagues compared shock-treated VT event burden and appropriate shock burden between the 2 study arms [2]. Shock-treated VT event burden was defined as the total number of VT events treated with ≥1 ICD or external shock. Appropriate shock burden was calculated as the total number of appropriate ICD or external shocks, regardless of the number of VT events.
The number of shock-treated VT events per 100 person-years was lower in the catheter ablation arm (39.07) than in the escalated AAD therapy arm (64.60; Anderson-Gill HR 0.60; 95% CI 0.38–0.95; see Figure). Similarly, the number of appropriate shock events favoured the ablation arm (48.35) over the AAD arm (78.23; Anderson-Gill HR 0.61; 95% CI 0.37–0.96).
Figure: Shock-treated ventricular tachycardia event burden [2]
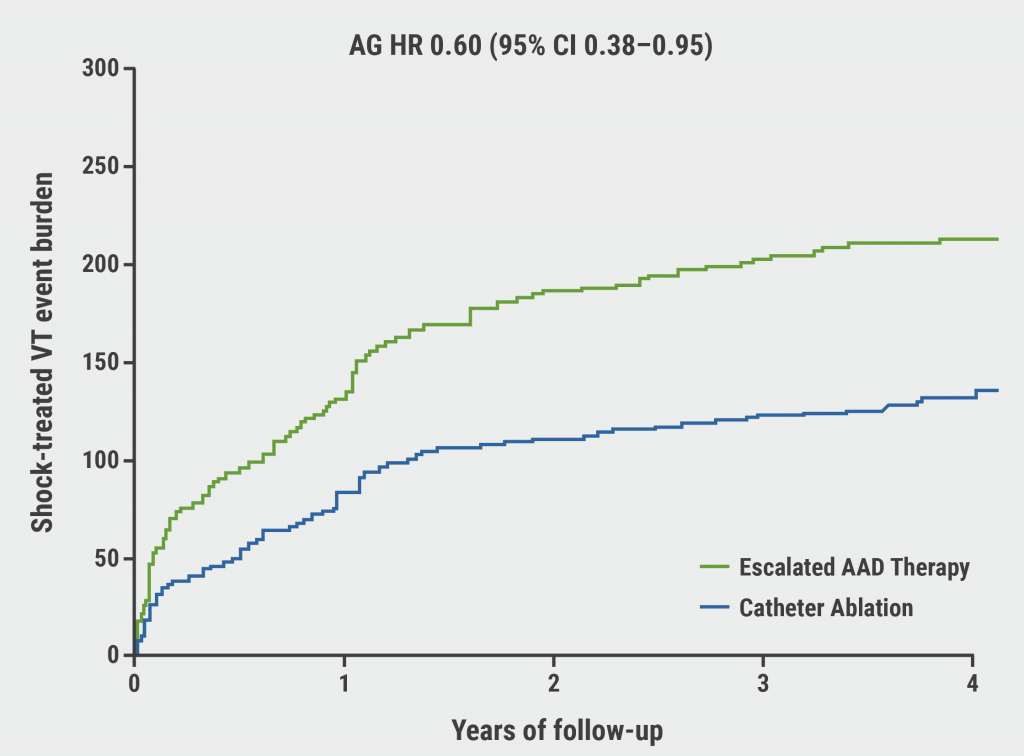
AAD, anti-arrhythmic drug; AG HR, Anderson-Gill hazard ratio; CI, confidence interval VT, ventricular tachycardia.
“Among patients with AAD-refractory VT and a prior MI, catheter ablation reduced shock-treated VT event burden by 40% and appropriate shock burden by 39%, compared with escalated AAD therapy,” concluded Dr Samuel.
- Sapp JL, et al. N Eng J Med 2016;375:111–121.
- Samuel M, et al. Reduction in shock burden with catheter ablation versus escalated antiarrhythmic drug therapy: Insights from the VANISH trial. News from ventricular ablation, EHRA 2022, 3–5 April, Copenhagen, Denmark.
Copyright ©2022 Medicom Medical Publishers
Posted on
Previous Article
« Low AF recurrence rates after PVI using pulsed-field ablation Next Article
Real-world safety results on pulsed-field ablation with pentaspline catheter »
« Low AF recurrence rates after PVI using pulsed-field ablation Next Article
Real-world safety results on pulsed-field ablation with pentaspline catheter »
Table of Contents: EHRA 2022
Featured articles
Letter from the Editor
Diagnostics and Prevention
Cardiac magnetic resonance imaging improves prediction of post-MI sudden cardiac death
AI model accurately predicts sudden cardiac death in overall population
AI model accurately discriminates between arrhythmias
Impact of AF screening on stroke prevention influenced by systolic blood pressure
Developments in Devices
Conduction system pacing potential alternative for biventricular pacing in heart failure
Left bundle branch area pacing is a feasible technique for HF and bradyarrhythmia
Focus on the efficacy of cardiac resynchronisation therapy in HF plus concomitant AF
RESET: No survival benefit of CRT-defibrillator over CRT-pacemaker in heart failure
Insertable cardiac monitors effective for AF detection in cryptogenic stroke
Updates on Ablation
First results of the POWER FAST III trial
Real-world safety results on pulsed-field ablation with pentaspline catheter
VANISH: Ablation reduces shock burden compared with anti-arrhythmic drug in ventricular tachycardia
Low AF recurrence rates after PVI using pulsed-field ablation
Pulsed-field ablation reduces neurocardiac damage versus cryoballoon ablation
Ultrasound-guided femoral venipuncture reduces complications in catheter ablation
News on Atrial Fibrillation
Sex differences revealed in AF determinants and AF progression
Early rhythm-control therapy efficacious in men and women with AF
Progression in remote app-based monitoring of atrial fibrillation
Other Topics
Benefits of SGLT2 inhibitors may extend beyond HF-associated outcomes
Related Articles
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy

