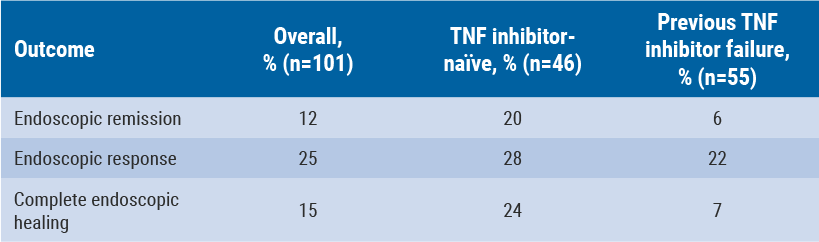
Although anti-TNF antibodies have been shown to be effective in many CD patients, a relevant subgroup does not respond to this therapy [14]. The underlying molecular mechanisms associated with endoscopic resistance to anti-TNF therapy were determined by Schmitt et al. [15].
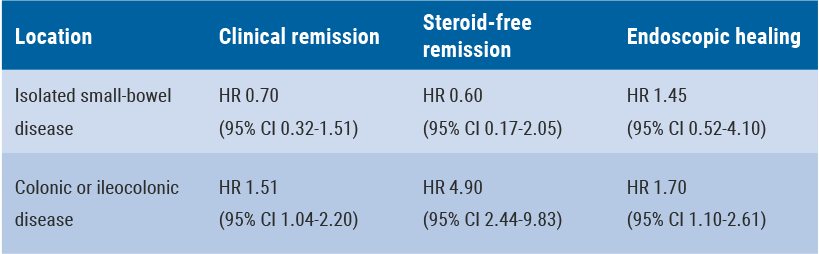
In this study, mucosal and blood cells from 197 CD patients were isolated prior to and during anti-TNF therapy. CD patients responding to anti-TNF therapy displayed a significantly higher expression of TNF receptor 2 (TNFR2) on mucosal T cells than non-responders prior to initiation. However, the expression of IL-23 receptor (IL23R) was not increased. Array analyses of differentiated gene regulation profiles in intestinal biopsies were compared in endoscopic non-responding CD patients and responders during ongoing anti-TNF therapy.
Within the cohort of CD susceptibility genes, there was a significant upregulation of genes that are associated with IL23R-dependent signalling pathways in anti-TNF non-responders. Apoptosis-resistant TNFR2+IL23R+ T cells were significantly more expanded in anti-TNF non-responders compared with responders and expressed the gut-tropic integrins α4β7. These cells exhibited increased expression of IFN-γ, T-bet, IL-17A and RORγt compared with TNFR2+IL23R- cells, indicating a mixed Th1/Th17-like phenotype. Intestinal TNFR2+IL23R+ T cells were activated by IL-23 derived from CD14+ macrophages, which were significantly more present in non-responders than responders prior to anti-TNF treatment. Administration of IL-23 to anti-TNF-treated mucosal organ cultures led to the expansion of CD4+IL23R+TNFR2+ lymphocytes. There was no accumulation of CD4+TNFR2+ T cells that were negative for IL23R.
Functional studies demonstrated that anti-TNF-induced apoptosis in mucosal T cells is abrogated by IL-23. It was concluded that expansion of apoptosis-resistant intestinal TNFR2+IL23R+ T cells is associated with resistance to anti-TNF therapy in CD. IL-23 is centrally involved in mediating resistance to anti-TNF therapy in CD patients, thereby representing a suitable molecular target in patients refractory to anti-TNF therapy.
- Wils P, et al. Clin Gastroenterol Hepatol. 2016;14:242-50.
- Schmitt H, et al. OP012. ECCO 2018.
Posted on
Previous Article
« Early and significant effects with upadacitinib induction treatment Next Article
Overall manageable safety profile of tofacitinib in long-term use »
« Early and significant effects with upadacitinib induction treatment Next Article
Overall manageable safety profile of tofacitinib in long-term use »
Table of Contents: ECCO 2018
Featured articles
IBD diagnostics
IBD disease patterns and genetics
Novel treatment strategies
Efficacy and safety of biologics
Oncology in IBD
Surgery for IBD
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy