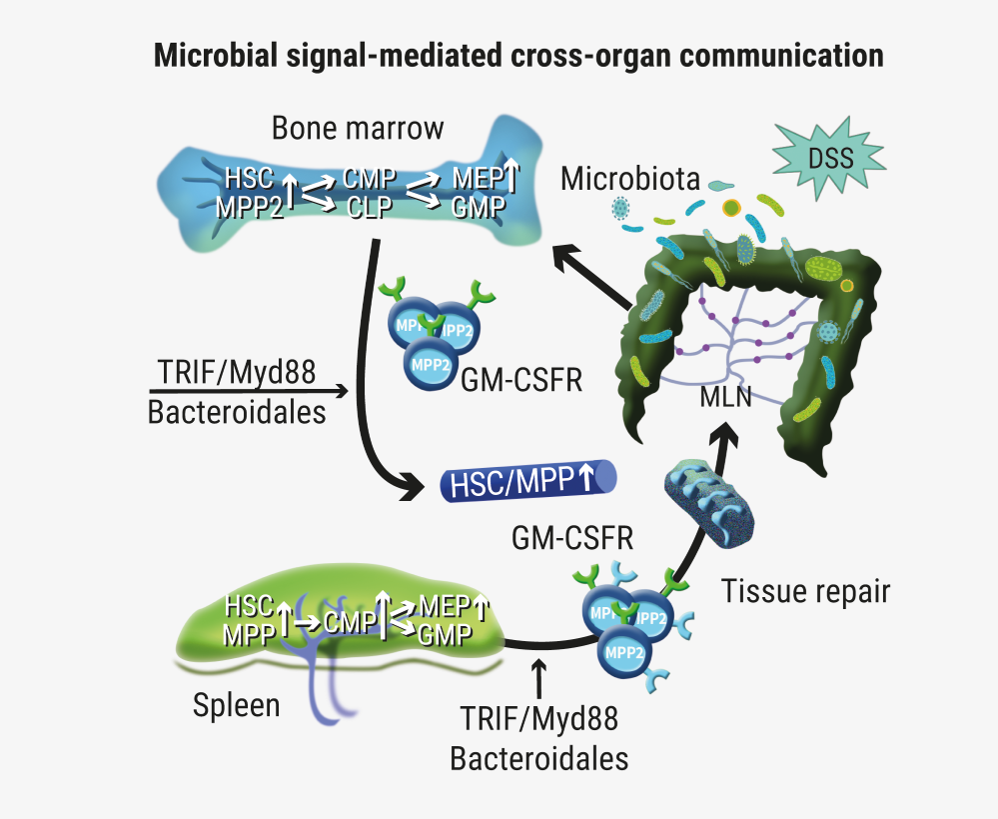
Haematopoietic stem cells (HSCs) sustain lifelong haematopoiesis, while lineage-restricted progenitors actively divide and mainly contribute to daily haematopoiesis. Haematopoietic challenges such as inflammation or infection activate HSCs to self-renew. Systemic challenge of gram-negative bacteria directly activates dormant HSCs to proliferate and impairs their competitive fitness via Toll-like receptor (TLR)-4 signalling [2]. To test their hypothesis that progenitor cells (HSPCs) can sense tissue damage signals derived from distal organs such as the gut, and translate that to haematopoietic production, Dr Takizawa’s team induced inflammatory bowel disease (IBD) in mice by adding dextran sodium sulfate (DSS) to their drinking water.
Inducing inflammatory bowel disease in the mice enhanced proliferation and expansion of HSPCs such as MPP and myeloid-restricted progenitors in bone marrow, while simultaneously reducing lymphoid-restricted progenitors, as measured by flow cytometry, microscopy, in vivo pharmacological treatment, and in vivo serial transplantation. Proliferating MPPs were localised adjacent to endothelial cells within the gut-associated mesenteric lymph node (MLN) but not in other lymph nodes, suggesting a specific haematopoietic response to gut inflammation, which the researchers demonstrated to be dependent on TLR signalling. Mice were then pre-treated with either a single or a mixture of antibiotics to deplete specific types of bacteria to determine what might trigger the TLR signalling. Pre-treatment with gram-positive directed antibiotics like vancomycin and ampicillin, completely abrogated haematopoietic responses to IBD, whereas neomycin and metronidazole, directed against gram-negative bacteria, enhanced haematopoietic response. Genome profiling and bacterial lysate injection identified that gram-negative bacterial species prompt MPP migration to the MLN through TLR-related signals. Following the MPP recruitment, myeloid cells including eosinophils and monocytes rapidly increased in MLN and their cell depletion by neutralising antibody worsened IBD-induced colitis.
This data suggests that in response to intestinal damage, bone marrow haematopoietic PCs sense microbiota via innate immune receptor and induce HSPC expansion followed by directional MPP migration to inflamed lymphoid tissues and generation of myeloid cells specialised for intestinal tissue repair (see Figure).
Figure: Schematic of the working hypothesis presented by Dr Takizawa et al [1]
- Takizawa H, et al. Abstract LB2604, 24th Congress of the EHA, 13-16 June 2019, Amsterdam, the Netherlands.
- Takizawa H, et al. Cell Stem Cell. 2017 Aug 3;21(2):225-240.e5
Posted on
Previous Article
« Infectious complications mild and not common in patients receiving CAR-T therapy for diffuse large B cell lymphoma Next Article
Overcoming the “don’t eat me” signal in AML and MDS »
« Infectious complications mild and not common in patients receiving CAR-T therapy for diffuse large B cell lymphoma Next Article
Overcoming the “don’t eat me” signal in AML and MDS »
Table of Contents: EHA 2019
Featured articles
Editor Biography
Interview with EHA President Prof. Pieter Sonneveld
Myeloid Malignancies
Residual disease in AML patients prior to stem cell transplant increases relapse risk
Gilteritinib prolongs overall survival in patients with FLT3-mutated relapsed/refractory AML
Initial data on AMV564 in patients with relapsed/refractory AML
Overcoming the “don’t eat me” signal in AML and MDS
Asciminib plus imatinib in patients with heavily pre-treated chronic myeloid leukaemia
Guadecitabine vs treatment of choice in AML
Lymphoid Malignancies
Unmutated IGHV as predictive factor for venetoclax/obinutuzumab benefit in frontline CLL
CAR-T cell therapy in ALL as breakthrough advance
Brentuximab vedotin continues to demonstrate superior clinical activity in classical Hodgkin lymphoma
Infectious complications mild and not common in patients receiving CAR-T therapy for diffuse large B cell lymphoma
Obinutuzumab/polatuzumab in follicular lymphoma
Exciting survival data for ibrutinib vs placebo in treatment-naïve, early-stage CLL
ASCEND study: Acalabrutinib improves progression-free survival in relapsed/refractory CLL
Venetoclax-obinutuzumab combination elicits high response rates in CLL
Myeloma
CASSIOPEIA trial: Phase 3 results of daratumumab + bortezomib/thalidomide/dexamethasone in multiple myeloma
Chimeric antigen receptor T cell therapy in multiple myeloma
Higher levels of treatment satisfaction without compromising efficacy: subcutaneous daratumumab in RRMM
Adding isatuximab to pomalidomide and dexamethasone improves PFS and ORR in RRMM
Subcutaneous daratumumab + cyclophosphamide, bortezomib, and dexamethasone in patients with newly diagnosed amyloid light chain amyloidosis
Venetoclax for multiple myeloma: effective but some safety concerns
Benign Haematology
New sickle cell drug voxelotor boosts levels of haemoglobin
Positive initial data evaluating the safety and efficacy of IMR-687 for treatment of sickle cell disease
Haematopoietic stem cell transplantation improves stroke risk in children with sickle cell anaemia
Early trial data shows positive results for treating anaemia in patients with end-stage renal failure
Bench-to-Bedside
Transformation of foetal haematopoietic stem and progenitor cells in the background of trisomy 21
Treating thalassemia twice, in mice
Haematopoietic stem cells can sense tissue damage in the gut
Promising news for gene therapy for sickle cell disease
Related Articles

August 9, 2019
CAR-T cell therapy in ALL as breakthrough advance
© 2024 Medicom Medical Publishers. All rights reserved. Terms and Conditions | Privacy Policy

